- Home
- News
- Spotlight on Science
- Tracing titanium...
Tracing titanium dioxide nanoparticles in the environment
03-01-2019
The abundance of titanium dioxide nanoparticles in the environment originating from human activities could be a potential environmental problem. To identify and distinguish between titanium dioxide nanoparticles from natural and anthropogenic sources, synchrotron techniques were used to study the nanoparticles in sewage sludge and soil.
Titanium dioxide nanoparticles are one of the most commonly produced nanomaterials worldwide. They are present in many consumer products, such as sunscreen and confectionery. After use, a large proportion of TiO2 ends up in agricultural soils, through the application of sewage sludge. A prerequisite for risk assessment is to be able to distinguish this released TiO2 from the elevated TiO2 natural background, and therefore detect anthropogenic inputs. Furthermore, the current models of nanomaterial fluxes in environmental compartments have large uncertainties, therefore field monitoring is needed to be able to quantify actual anthropogenic inputs. Therefore, an evaluation of the differences in terms of composition and structure between natural and anthropogenic nanomaterials may provide clues to help interpret data on toxicity.
In this study, the potential of physical techniques to distinguish natural versus anthropogenic particles has been investigated. Three matrices were compared: sewage sludge, agricultural soil that had never received sewage sludge, and sludge-amended soil. They were studied using micro and nano X-ray fluorescence and bulk and micro X-ray absorption spectroscopy at beamlines ID21 and ID16B, by X-ray diffraction at beamline BM25, and by transmission electron microscopy.
The size distribution of TiO2 particles was determined by image analysis of the Ti nanoXRF maps. This revealed that the soil and sludge have a similar size distribution, with a range of nano- and micro-scale particles. Bulk Ti K-edge XANES spectroscopy showed that the two major Ti species were the TiO2 polymorphs anatase and rutile. In the sludge, there were roughly equal proportions of rutile and anatase. This information is important because rutile and anatase have different toxicities, and it is very difficult to know which polymorph is used in which type of product, and the global proportions of rutile and anatase released into the environment. Amorphous TiO2 was also detected as a minor species in the soil and in the sludge by µXANES. The combination of µXRF and µXANES data showed that there was no relationship between the size of the aggregates and Ti speciation.
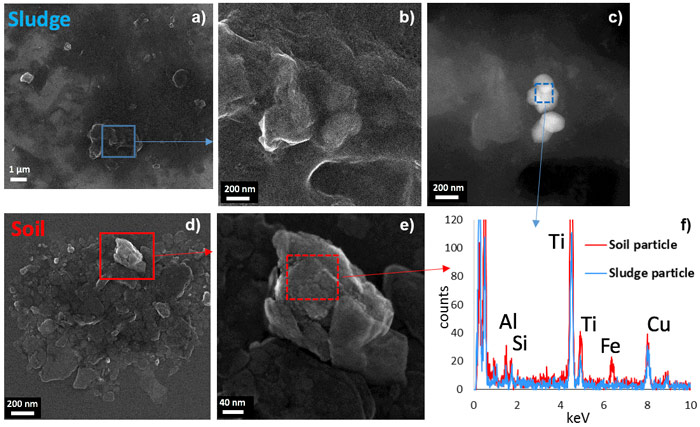
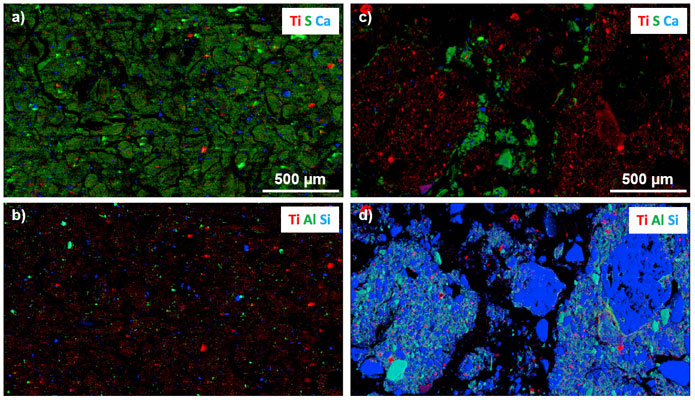
Finally, the morphology of the crystals observed by electron microscopy and the status of TiO2 particles within the organo-mineral aggregates proved to be relevant criteria to discriminate natural versus anthropogenic TiO2. In the sludge, smooth particles typical of TiO2 pigments were evidenced (Figure 1c, 1f), whereas the soil contained rough and irregular TiO2 particles. Moreover, TiO2 particles in the sludge were present as weakly evolved aggregates, dominated by organic matter (Figure 1a-b and Figure 2), whereas in the soil they were intimately associated with organo-mineral assemblages forming the soil micro- and macroaggregates (Figure 1d-f and Figure 2), the building blocks of soils.
 |
|
Figure 1. TEM analyses of the TiO2 particles present in sludge (a–c) and soil (d–e). (f) Energy-dispersive X-ray microanalysis of the zones highlighted in (c) and (e). |
Although TiO2 phases are generally considered as very weakly soluble, they undergo weathering and transformations in acidic soils or in the rhizosphere [1]. Thus, the observed differences in particle morphology may attenuate over time due to the weathering of TiO2 minerals. Likewise, even if it is slow (in the range of decades, although this has been debated), the formation of soil aggregates is a dynamic and continuous process, so the progressive incorporation of anthropogenic TiO2 within soil organo-mineral assemblages is expected. It is likely that with time, engineered TiO2 becomes indistinguishable from the natural background in soils.
 |
|
Figure 2. Tricolour μXRF maps for sewage sludge (a,b) and for sludge-amended soil (c,d), showing the distribution of Ti, S, Ca, Al and Si. |
To conclude, the X-ray studies complemented microscopy methods providing chemical identification of the TiO2 species and location of the nanoparticles within soil and sludge matrixes.
Principal publication and authors
Searching for relevant criteria to distinguish natural vs. anthropogenic TiO2 nanoparticles in soils, A.E. Pradas del Real (a,b), H. Castillo-Michel (b), R. Kaegi (c), C. Larue (d), W. de Nolf (b), J. Reyes-Herrera (b), R. Tucoulou (b), N. Findling (a), E. Salas-Colera (b) and G. Sarret (a), Environ. Sci.: Nano 5, 2853- 2863 (2018); doi: 10.1039/c8en00386f.
(a) ISTerre (Institut des Sciences de la Terre), Univ. Grenoble Alpes, CNRS, Grenoble (France)
(b) ESRF
(c) Eawag, Particle Laboratory, Dübendorf (Switzerland)
(d) ECOLAB, Universite de Toulouse, CNRS, INPT, UPS, Toulouse (France)
References
[1] M. Schindler and M.F. Hochella, Geology, 44, 515–518 (2016).



