- Home
- News
- General News
- How cadmium in algae...
How cadmium in algae alters the carbon cycle
21-11-2019
Plants in water use carbon dioxide, but scientists have found and quantified, using the ESRF, that toxic metals such as cadmium can alter the carbon dioxide fixation on the algae, contributing in this way to the unbalance of the carbon cycle. Their results are published in Plant, Cell and Environment.
The increase of carbon dioxide concentration in the atmosphere over the past decades has been associated with global climate change. Therefore, investigation and understanding of the biogeochemical carbon cycle is of primordial importance. The terrestrial and oceanic biosphere plays a major role in the global carbon cycle by the production and conversion of organic matter. For the first time, scientists have managed to assess the impact of cadmium in the carbon assimilation in microalgae.
Plants have developed a process for CO2 fixation into organic carbon using the enzyme ribulose-1,5-bisphosphate carboxylase/oxygenase (RuBisCO), which is considered the most abundant protein in the biosphere. Microalgae are small eukaryotic organisms where RuBisCO is concentrated in the specific organelle called the pyrenoid. Pyrenoid-containing algae are responsible for 28 - 44% of the global carbon fixation, so microalgae play a key role in the biogeochemical carbon cycle.
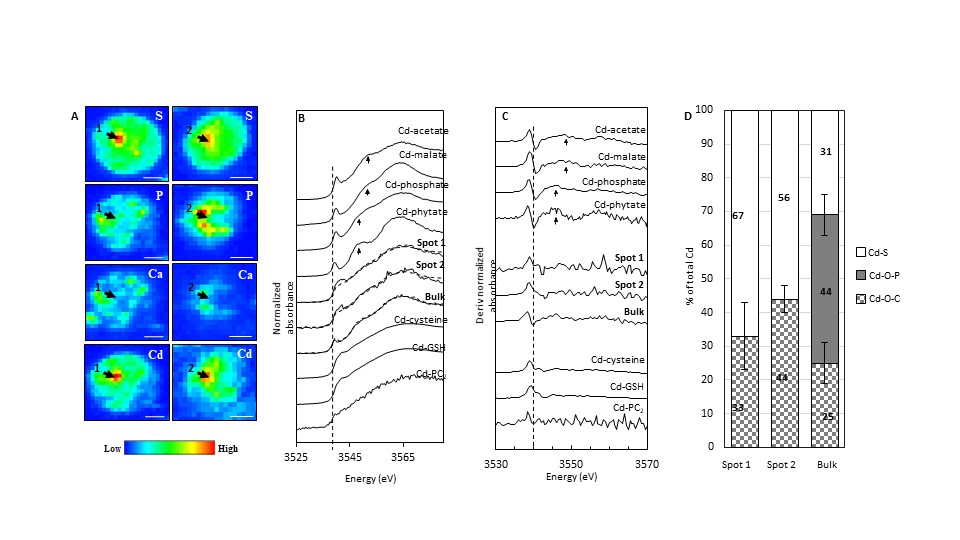
A team of scientists from the CNRS, Université de Pau, Martin-Luther-Universität Halle-Wittenberg, Université de Bordeaux and the ESRF studied a unicellular green alga found worldwide in soils and fresh water, called Chlamydomonas reinhardtii, using X-ray beam imaging techniques and Nanoscale secondary ion mass spectrometry (NanoSIMS). X-ray fluorescence and X-ray absorption near-Edge at the ESRF indicated the localization and speciation of cadmium in the cellular structure. “Our work at ESRF was highly important to show that cadmium binds in the algae to the enzyme RuBisCO and thus hinders CO2 fixation”, says Dirk Schaumlöffel, corresponding author of the paper.
 |
|
In situ cadmium localization and speciation in C. reinhardtii exposed to 70 μM Cd for 48 in TAPEDDHA medium. (A) False color μ-XRF elemental maps of Cd, S, P and Ca, arrows show points of interest where μ-XANES analyses were performed, scale bar = 2 μm. Step size = 0.4 μm, dwell-time = 300 ms/pixel at 3570 eV for S, P, Cd and at 4100 eV for Ca. (B) Two or three components fits (dotted line) of Cd LIII-edge μ-XANES spectra (solid line) of points of interest marked by arrows and of bulk sample; (C) Derivative of Cd LIII-edge μXANES; (D) Distribution of Cd ligands after normalization of the percentages to 100%. |
The results showed that cadmium clearly impairs carbon assimilation in C. reinhardtii grown in mixotrophic conditions. Mixotrophy means, in this case, that the algae use not only CO2 but also other carbon sources such as organic acids. Due to the pyrenoidal sequestration of cadmium, CO2 fixation is limited during cadmium exposure. Mixotrophy could therefore allow phytoplankton surviving toxic metal pollution events. Although mixotrophy seems to render aquatic ecosystems more resistant, the alteration of CO2 fixation by toxic metals could contribute to the unbalance of the biogeochemical carbon cycle.
The next step for the team is to investigate the influence of other toxic metals and reveal the metabolic pathway under metal stress.
Reference:
Penen F. et al., Plant, Cell and Environment, 2019, accepted. https://doi.org/10.1111/pce.13674 DOI: 10.1111/pce.13674
Text by Montserrat Capellas Espuny
Top image: Algae in a lake.



