- Home
- News
- General News
- Bringing cryo-correlative...
Bringing cryo-correlative hard X-ray microscopy to life science
27-02-2026
Scientists led by the ESRF, UGA and INSERM have developed cryo-correlative nano-imaging, a new technique that combines lab cryo-fluorescence microscopy, cryo X-ray fluorescence nanoimaging and phase-contrast nano-tomography on ID16A. The results are published in ACS Nano.
Share
Biologists have long wanted to answer a deceptively simple question: what are the structures we see inside cells actually made of? Visible light fluorescence microscopy shows where organelles are, but not their chemical composition. Hard X-rays can map the chemistry but do not necessarily see the organelles. Cryo-correlative nanoprobe work remains rare, particularly for 3D elemental imaging of whole frozen cells.
A new study at ID16A beamline of the ESRF offers a practical solution. An international team has developed an integrated cryogenic workflow that links laboratory cryo-fluorescence microscopy to targeted cryo X-ray fluorescence (XRF) nano-imaging and phase-contrast nano-tomography.
With this new method, they have tracked therapeutic nanoparticles from the European ScanNtreat project as they moved through cancer cells, showing both where the particles went and what happened to them.
The first author of the publication, Dmitry Karpov, former ESRF scientist and now researcher at the Université Grenoble Alpes, explains how this new development can lead to applications: “This is an example of what the ESRF aims to do: to turn cutting-edge instrumentation into discoveries with direct impact on people's lives, in this case for medicine and life sciences”.
The key is in the ice
The challenge is cell preservation. Diffusible ions and trace elements in the cell can be distorted by dehydration, chemical fixation, or radiation damage. To overcome this, the new workflow keeps cells vitrified (flash frozen to prevent ice crystals) and maintains them well below the devitrification point during transfers and measurements.
Efficiency matters too. Synchrotron time is precious, so cells are prescreened using a lab cryo-fluorescence system (shared by Inserm UA7 and ESRF) to find promising targets. Only those make it to the nanoprobe.
Painting organelles with heavy atoms
Fluorescence microscopy is great for spotting labeled organelles but tells us little about their quantitative chemistry. Nano-XRF does the opposite: it measures many elements at once, but organelles and cytoplasm often look identical in hard X-rays because their native elemental profiles are similar.
The research team bridged this gap with heavy-atom molecular probes (halogens and transition/lanthanide metals) developed by collaborators at Curtin University in Australia. These act as "XRF paint," lighting up specific organelles without requiring different sample preparation for the two imaging modes. The heavy atoms are part of bio-orthogonal molecules, so they neither interfere with the workings of the cells nor get confused with elements native to the cell. The result: correlative images built entirely from X-ray fluorescence.
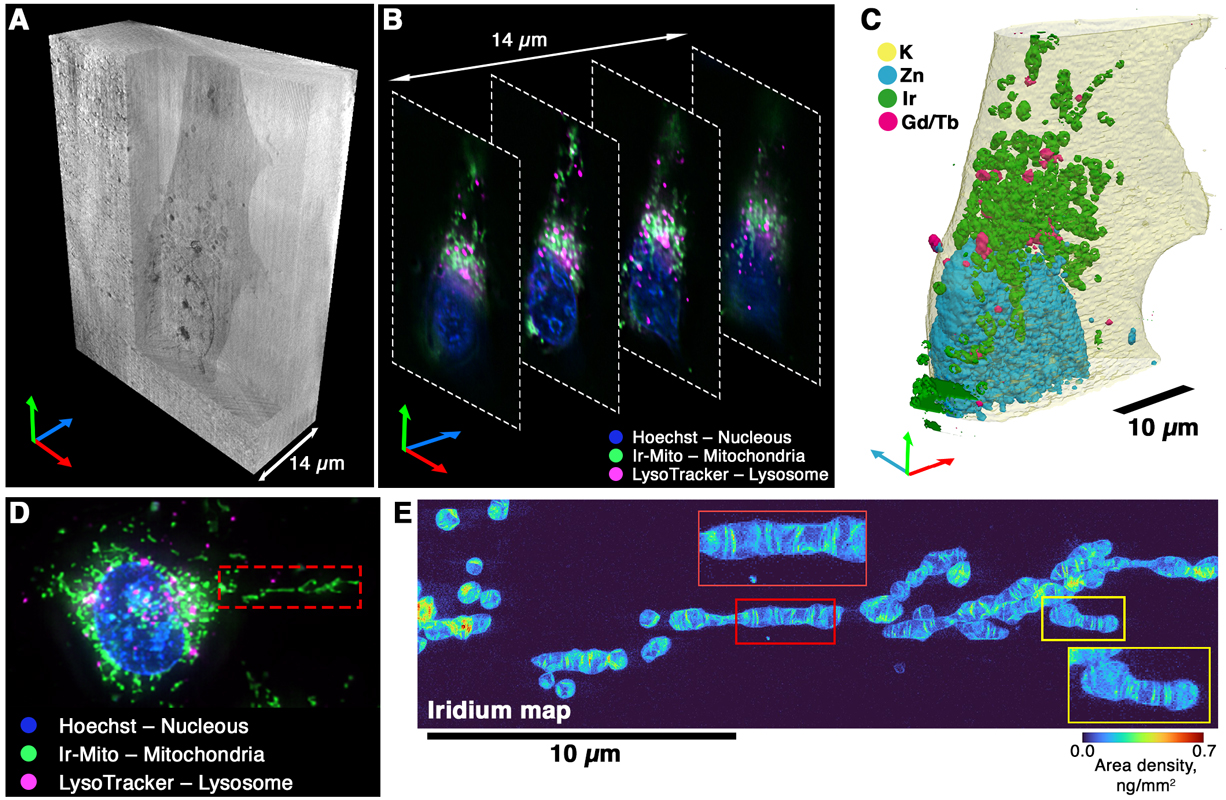 |
|
Three-dimensional visualization of the cellular structures with the proposed cryo-correlative workflow. (A) Volume rendering of the phase contrast nano tomography data. (B) Slices through the cryo-optical fluorescence microscopy data. (C) Volume rendering of the elemental distribution obtained with X-ray fluorescence nano tomography. (D) Cryo-FLM image of another cell highlighting an extended mitochondrial structure, outlined by a red dashed rectangle for higher-resolution cryo-nano-XRF scanning. (E) XRF map of Ir acquired at a 30 nm pixel size. Red and yellow rectangles mark the magnified regions of interest. Credits: Karpov, D., et al, ACS Nano. |
Tracking therapeutic nanoparticles
To show translational relevance, the team investigated pegylated lanthanide nanohybrids (gadolinium and terbium fluoride nanoparticles from ScanNtreat European project) inside frozen triple-negative breast cancer cells. The gadolinium and terbium signals localised in lysosome-rich regions, consistent with uptake and sequestration pathways that can now be addressed more directly using this correlative workflow.
Because XRF is quantitative, the team could go beyond qualitative observation. The publication reports lysosomes containing up to 15 femtograms of gadolinium and 2 femtograms of terbium, roughly 60 nanoparticles per lysosome under these conditions. That compartment-level detail is precisely what is needed to understand nanoparticle integrity, trafficking and intracellular fate, all of which are critical questions in nanomedicine development.
A nanoscale look inside mitochondria
The same workflow, pushed to 20–30 nm resolution on ESRF's ID16A nanoprobe, revealed something unexpected. An iridium complex developed at Adelaide University labels mitochondria, and the iridium signal in cryo-nano-XRF maps matched beautifully. But it also showed quasi-periodic striations inside the mitochondria.
At 30 nm pixel spacing, line profiles picked up repeating patterns every 60–150 nm – consistent with the high-curvature folds of the inner mitochondrial membrane (cristae) reported by super-resolution light, soft X-ray and electron microscopy techniques. The organelle tag wasn't just marking location; it was enhancing ultrastructure in the elemental data.
What comes next
Cryo-nano-XRF tomography is currently the only way to map elemental distributions in 3D across a whole frozen cell at true nanoscale, but only a handful of beamlines can do it routinely. That makes continued development of cryo-correlative hard-X-ray workflows both timely and strategic.
“This work is the culmination of many years of developing this technique”, explains Sylvain Bohic, INSERM scientist and scientific collaborator at the ESRF and corresponding author of the publication. “We have identified the needs of researchers and thanks to the work of our interdisciplinary team, we can now offer a solution”.
The study lays out concrete next steps: higher detector throughput (multi-element arrays like ARDESIA-16), better stability and metrology, smarter dose-aware reconstructions denoising and ultimately a new beamline ID18 that is optimized for high through-put and automation. “Our goal is full-cell 3D XRF nanotomography at 50 nm resolution in a reasonable amount of time and it is starting to look achievable”, concludes Peter Cloetens, scientist in charge of ID16A.
Reference:
Karpov, D. et al, ACS Nano, ASAP article, 14 February 2026. https://doi.org/10.1021/acsnano.5c10637



