- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2016
- Structural biology
- Influenza polymerase can adopt an alternative configuration involving a radical repacking of PB2 domains
Influenza polymerase can adopt an alternative configuration involving a radical repacking of PB2 domains
Structural studies using X-ray crystallography and SAXS reveal a second configuration of influenza polymerase in which flexibly-linked domains, peripheral to the core of the polymerase, repack in a completely different way. This state is transcriptionally inactive and may correspond to an intermediate in the assembly of the newly synthesised polymerase with the viral RNA.
Influenza virus causes widespread disease each year and novel influenza strains have the potential to provoke dangerous pandemics as last happened in 2009. The key enzyme for the multiplication of the virus in infected cells is the viral RNA-dependent RNA polymerase. This is a heterotrimeric, multi-functional machine with subunits denoted PA, PB1 and PB2, which uses the viral RNA genome as template for both (a) transcription into viral mRNA coding for viral proteins and (b) replication to generate new genome copies that are eventually packaged into progeny virions. Because of its critical role in the viral life cycle, the influenza polymerase is being actively pursued as a target for novel anti-viral drugs and thus detailed structural information on the various functional states of the polymerase is highly desirable.
 |
|
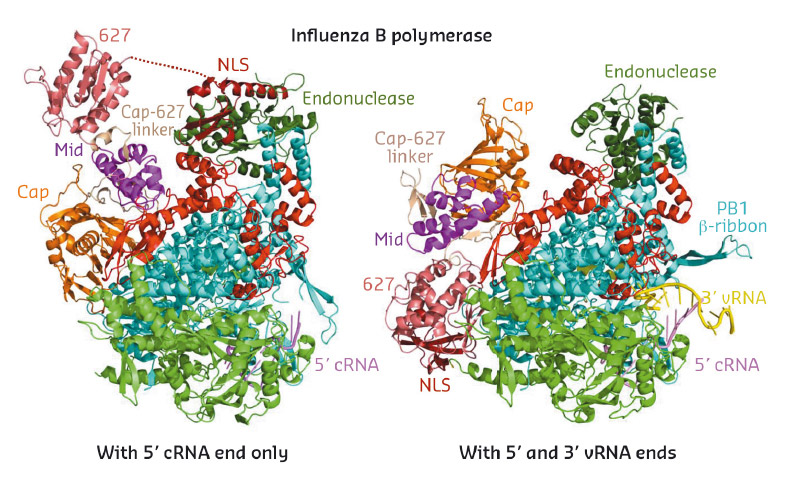
Fig. 76: Ribbon diagrams comparing the crystal structures of the new conformation of influenza B polymerase (left, 5' cRNA bound) to the previous published conformation (right, full promoter bound). The polymerase core comprises PA-C (green), PB1 (cyan) and PB2-N (red). The flexibly linked peripheral domains are individually labelled. |
In 2014, the crystal structure of the complete influenza A and B polymerases bound to their so-called promoter (the 3' and 5' extremities of the viral RNA) were determined [1,2]. This work led to a structure-based model that explained how the polymerase performs the first step of transcription by a unique mechanism known as 'cap-snatching' [2], whereby nascent host-cell transcripts are pirated to provide short capped primers to kick-start transcription of viral mRNAs. In the follow up work described here, the influenza B polymerase was crystallised without the complete promoter in an unexpected and remarkably very different conformation. Whereas the core of the polymerase, comprising the C-terminal two-thirds of the PA subunit (PA-C), the PB1 subunit and the N-terminal third of the PB2 subunit (PB2-N), does not significantly change, the peripheral domains of PA-N (the cap-snatching endonuclease) and PB2-C (the cap-binding, mid-link, 627 and NLS domains), which are joined together or to the core by flexible linkers, radically change their position, orientation and packing arrangement (Figures 76 and 77). As an example, the small C-terminal PB2 NLS (nuclear localisation signal) domain translates a remarkable 93 Å and in the new structure makes completely novel interactions with the endonuclease domain (Figure 76, left). Additional high resolution crystal structures of the PB2 NLS-endonuclease interaction and of the PB2-C cap-binding, mid-link, 627 domains, which showed the same inter-domain arrangements as in the new complete polymerase structure, confirm that the observed configuration is unlikely to be a crystal packing artefact.
 |
|
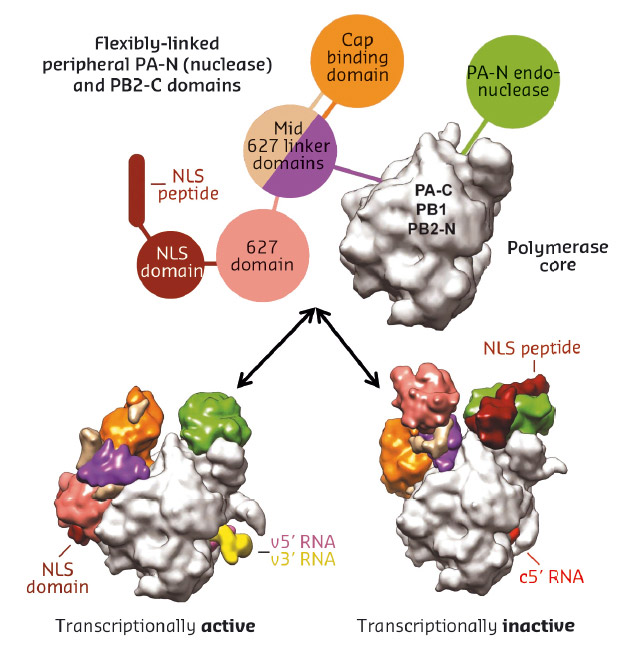
Fig. 77: Schematic diagram illustrating the rearrangement of peripheral domains in the two crystallised states of influenza B polymerase. |
To complement our crystallographic studies, small-angle X-ray scattering (SAXS) and cross-link mass-spectrometry (XL-MS) studies were performed in solution. The SAXS measurements, carried out at beamline BM29, clearly showed that when no promoter is bound or just the 3’ end of the vRNA is bound, the polymerase is in a much more open conformation (i.e. larger radius of gyration) than when the 5’ end or the full promoter is bound. Moreover, the XL-MS results could only be explained by the polymerase interchanging between a number of configurations, probably more than the two distinct arrangements observed in crystal structures in solution. Finally, biochemical studies showed that ‘cap-snatching’ as assayed by endonuclease cleavage of a capped RNA, was far more efficient when the full promoter was bound than when no vRNA was bound, correlating with the difference in compaction of the corresponding structures observed by SAXS.
We do not yet know to which functional state the new influenza polymerase configuration corresponds. However, there is good structural and biochemical evidence that the previously observed conformation with the full-promoter bound corresponds to the transcriptionally active state (Figure 77 bottom left). In contrast, the newly observed configuration, which has also been observed with Influenza C polymerase with no vRNA bound [3], appears to be unsuitable for cap-snatching, due to an inappropriate alignment of the cap-binding and endonuclease domains (Figure 77 bottom right). Instead this conformation may be an assembly intermediate in the transition from unbound to full promoter bound polymerase.
Principal publication and authors
Influenza polymerase can adopt an alternative configuration involving a radical repacking of PB2 domains, E. Thierry (a), D. Guilligay (a), J. Kosinski (b), T. Bock (b), S. Gaudon (a), A. Round (a), A. Pflug (a), N. Hengrung (c,d), K. El Omari (d), F. Baudin (a,b), D.J. Hart (a), M. Beck (b) and S. Cusack (a), Molecular Cell 61, 125–137 (2016); doi: 10.1016/j.molcel.2015.11.016.
(a) European Molecular Biology Laboratory Grenoble Outstation and Unit of Virus Host-Cell Interactions, University Grenoble Alpes-CNRS-EMBL Grenoble (France)
(b) Structural and Computational Biology Unit, European Molecular Biology Laboratory, Heidelberg (Germany)
(c) Sir William Dunn School of Pathology, University of Oxford (UK)
(d) Division of Structural Biology, Henry Wellcome Building for Genomic Medicine, University of Oxford (UK)
References
[1] A. Pflug et al., Nature 516, 355-360 (2014).
[2] S. Reich et al., Nature 516, 361-366 (2014).
[3] N. Hengrung et al., Nature 527, 114-117 (2015).



