- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2016
- X-ray nanoprobe
- The intracellular dissolution of silver nanoparticles reveals the mechanism of their toxicity
The intracellular dissolution of silver nanoparticles reveals the mechanism of their toxicity
Despite their toxicity to eukaryotic cells, silver nanoparticles are included in commercial products for their biocide activity through silver ion release. Silver nanoparticles intracellular dissolution was visualised and measured in hepatocytes, and the complexes formed by silver ions in cellulo were investigated.
Silver nanoparticles (AgNPs) are extensively used in healthcare products, fabrics, and food packaging for their antibacterial properties. Humans are therefore exposed to AgNPs, and the toxicity of AgNPs to eukaryotic cells is today a matter of concern. AgNPs can enter eukaryotic cells and the toxic effects they exert are most probably a consequence of Ag+ release from the AgNPs surface in cellulo. However, quantitative information concerning AgNP intracellular dissolution is missing and the mechanisms triggered by Ag+ poisoning are still debated. Dissolution rates in vitro depend on a variety of parameters such as particle size and coating, or pH and chemical composition of the medium. Although predicting the transformations that occur in complex biological systems is extremely challenging, it is pivotal in view of the design of safer NPs.
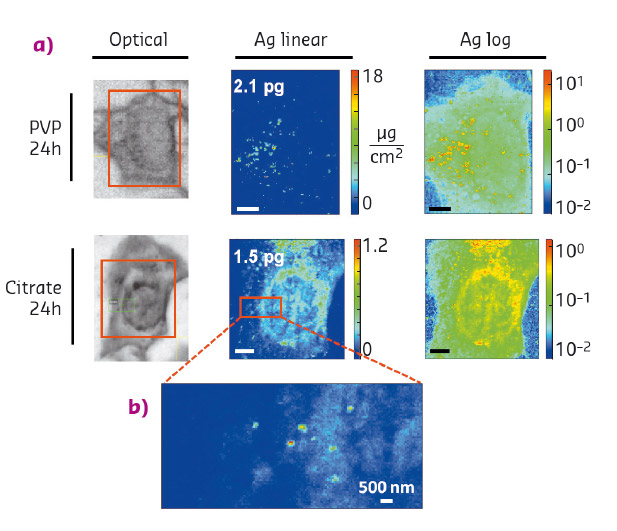
This study made use of the high-resolution X-ray fluorescence (XRF) imaging capabilities of beamline ID16B to map silver distribution in individual hepatocytes (HepG2 cells) exposed to AgNPs, with the aim of measuring the metal content as a function of the exposure time and to highlight the influence of particle coating on intracellular dissolution. We studied AgNPs with coatings polyvinylpyrrolidone (PVP) and citrate, which are commonly used in industrial AgNP formulations. Figure 44a shows silver distribution in hepatocytes exposed for 24 hours to AgNPs. Citrate-coated AgNPs dissolve more than the PVP-coated AgNPs at equal exposure time, although the total silver uptake is similar for the two formulations; this explains the higher toxicity of the citrate-coated NPs. High-resolution (70×70 nm2) XRF maps (Figure 44b) allow the visualisation of Ag hotspots of a few hundreds of nm in size, which correspond to endocytic vesicles containing NPs (as confirmed by TEM observations), whereas the weaker signal diffused throughout the cell corresponds to Ag+ species. The ionic component is not detected in TEM, but only in the nanoprobe, thanks to its attogram sensitivity.
 |
|
Fig. 44: Intracellular dissolution of AgNPs visualised by XRF imaging. a) Silver distribution in hepatocytes exposed to PVP- or citrate-coated AgNPs for 24 h. The areas of XRF hyperspectral images are highlighted in red over the optical micrographs. The values in pg are the total amounts of Ag in the map areas. Scale bars = 5 μm. b) High-resolution silver distribution in the area highlighted in panel (a). |
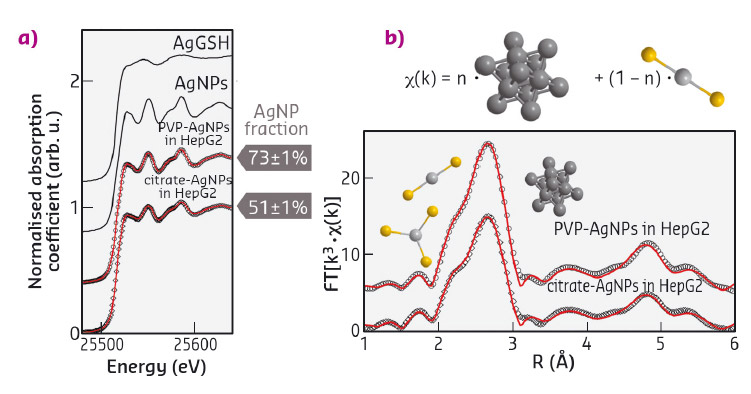
The fate of the Ag+ ions needs to be unravelled to obtain a deeper insight into the toxicity mechanisms. We have previously shown that Ag K-edge X-ray absorption spectroscopy (XAS) under cryogenic conditions can be used to measure the average dissolution of AgNPs in cultured cells, and to disclose the complexes formed in cellulo by the released ions [1]. The XANES spectra of hepatocytes exposed to AgNPs (Figure 45a), collected at beamline BM30B (FAME CRG), reveal that, after 24 h exposure, citrate-coated AgNPs released ~50% of their atomic content, while PVP-coated released only ~30%, confirming single-cell XRF observations. In both cases Ag+ is found to form complexes with thiolate (R-S–).
The binding properties of Ag+ are similar to those of Cu+, a physiological metal that preferentially binds to thiolates in vivo. Ag+ can therefore replace Cu+ in its native protein binding sites and induce the impairment of metal homeostasis. The ab initio analysis of Ag K-edge EXAFS spectra (Figure 45b) reveals the coordination chemistry of Ag+ in the Ag-thiolate complexes formed upon AgNP intravesicular dissolution. A coexistence of digonal AgS2 and trigonal AgS3 complexes is found, with an average Ag-S distance of 2.45 Å.
 |
|
Fig. 45: Ag K-edge (a) XANES and (b) EXAFS spectra of hepatocytes exposed to AgNPs for 24 h. Open symbols represent experimental data, red traces are best fit curves. XANES data (a) are fitted as linear combinations of reference compounds (black lines), EXAFS with the ab initio model depicted in (b). |
According to a previous study on the complexes formed by Ag+ with biomolecules involved in Cu homeostasis [2], the AgS2 and AgS3 complexes found in hepatocytes are most likely silver-glutathione and silver-metallothionein, respectively. An important overexpression of the genes of the metallothionein and of the enzyme involved in glutathione synthesis was observed in cells exposed to AgNPs, confirming that these two biomolecules are the major chelators of free intracellular Ag+.
The single-cell elemental information obtained through nano-XRF imaging combined with the averaged chemical information provided by XAS has revealed the transformations of metallic nanoparticles in a cellular model, and brought novel insight into the toxicity mechanisms.
Principal publication and authors
Visualization, quantification and coordination of Ag+ ions released from silver nanoparticles in hepatocytes, G. Veronesi (a,b), A. Deniaud (a), T. Gallon (a), P.-H. Jouneau (c), J. Villanova (b), P. Delangle (d), M. Carrière (d), I. Kieffer (e), P. Charbonnier (a) and E. Mintz (a), I. Michaud-Soret (a), Nanoscale 8, 17012-17021 (2016). doi: 10.1039/c6nr04381j.
(a) CNRS/CEA/UGA LCBM, Grenoble (France)
(b) ESRF
(c) CEA/UGA INAC/MEM, Grenoble (France)
(d) CEA/UGA/CNRS, INAC/SyMMES/CIBEST, Grenoble (France)
(e) BM30B/FAME, ESRF, Grenoble (France)
References
[1] G. Veronesi et al., Nanoscale 7, 7323-7330 (2015).
[2] G. Veronesi et al., Inorg. Chem. 54, 11688-11696 (2015).



