- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2007
- Materials Science
- Ultrafast diffraction captures a complex photo-dissociation process for CBr4 in solution
Ultrafast diffraction captures a complex photo-dissociation process for CBr4 in solution
The photochemistry of bromine-containing molecules is important for the composition of the atmosphere. When exposed to UV light from the sun, they can decay into free Br atoms which may react with ozone (O3) leading to a reduction in the ozone layer which protects living organisms from excessive solar UV exposure. Carbon tetrahalides such as CBr4 are found in the atmosphere and they constitute an important source of reactive halogens. Their photochemistry in the gas and condensed phase has therefore become an area of active investigation [1].
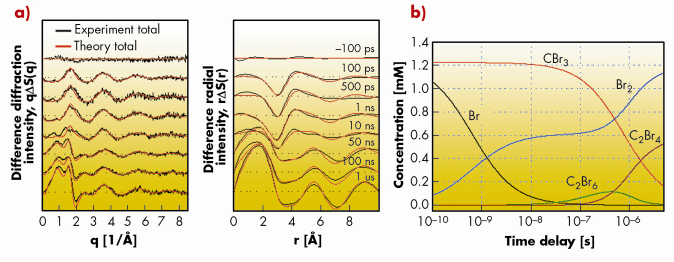
Studies of the photodissociation of tetrabromomethane by time-resolved optical and X-ray absorption spectroscopy have led to contradictory results in the past. None gave a complete and satisfactory description. Therefore the photochemistry of CBr4 in solution was investigated by time-resolved X-ray diffraction. This method offers the advantage that the scattered X-rays probe all interatomic distances in the sample, implying that all reaction intermediates are probed to within the experimental resolution. This is in stark contrast to optical spectroscopy where excited states might be optically silent, i.e. they would escape detection. Hence time-resolved X-ray diffraction offers an overall view of all intermediates. The laser pump X-ray probe setup on ID09TR allowed us to follow the structure and composition of the photochemical pathway of the excited CBr4 molecules in solution. The dissociation is triggered by the absorption of an ultraviolet 266 nm laser pulse (2 ps) which excites and breaks one of the four C-Br bonds and the ensuing structures are probed by a delayed 100 ps X-ray pulse. The experiment used a flowing sample and the pump-probe sequence was repeated at 986.3 Hz while the diffracted signal was accumulated on a MarCCD detector [2]. The difference diffraction signal of CBr4 in methanol at various time delays (Figure 39a) contains the dynamics of the reaction intermediates as a function of time.
 |
|
Fig. 39: Time-resolved difference diffraction signal of CBr4 in methanol as a function of time delays. (a) Difference diffraction intensities, q |
By modelling the measured diffraction intensities ![]() S(q, t) with putative density functional theory (DFT) structures, modelled in solution by Molecular Dynamics(MD), the concentration of the actual reaction intermediates can be determined (Figure 39b). The results show that the CBr3 and Br concentrations are dominant during the early stages, indicating the rupture of the C-Br bond in CBr4 by the UV excitation. The Br radical decays rapidly into Br2 by non-geminate recombination. The main reaction of CBr3 is to leave the solvation cage and to pair up with another CBr3 to form C2Br6. The C2Br6 intermediate is not stable and dissociates in microseconds to Br2 and a new species, C2Br4, as the final products. At longer time delays, the stable bromine and C2Br4 molecules become the dominant species. Br2 is formed through non-geminate recombination of Br at early time delays, whereas later, it is also formed from dissociation of C2Br6.
S(q, t) with putative density functional theory (DFT) structures, modelled in solution by Molecular Dynamics(MD), the concentration of the actual reaction intermediates can be determined (Figure 39b). The results show that the CBr3 and Br concentrations are dominant during the early stages, indicating the rupture of the C-Br bond in CBr4 by the UV excitation. The Br radical decays rapidly into Br2 by non-geminate recombination. The main reaction of CBr3 is to leave the solvation cage and to pair up with another CBr3 to form C2Br6. The C2Br6 intermediate is not stable and dissociates in microseconds to Br2 and a new species, C2Br4, as the final products. At longer time delays, the stable bromine and C2Br4 molecules become the dominant species. Br2 is formed through non-geminate recombination of Br at early time delays, whereas later, it is also formed from dissociation of C2Br6.
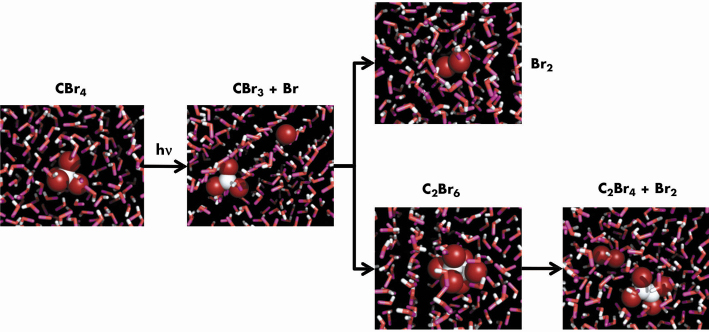
In conclusion, time-resolved X-ray diffraction has captured the complicated photodissociation pathways of tetrabromomethane in methanol over times ranging from 100 ps to 5 µs. X-ray diffraction offers the important advantage that the structure of short-lived and stable molecules are determined in one experiment: CBr3, Br, C2Br6, C2Br4 and Br2 and the associated rate constants connecting the complex reaction pathways were determined to ~ 1 pm spatial resolution and ~100 ps time resolution (Figure 40). Our experiment and analysis show that Br2 can be released from the photolysis of tetrabromomethane in the liquid phase indicating that polybromomethanes are potentially important sources of active bromine that destroys ozone in the atmosphere.
 |
|
Fig. 40: A schematic photochemical reaction mechanism of CBr4 in methanol determined by time-resolved X-ray diffraction in solution. |
References
[1] R. Vogt, P.J. Crutzen and R. Sander, Nature 383, 327 (1996).
[2] H. Ihee, M. Lorenc, T.K. Kim, Q.Y. Kong, M. Cammarata, J.H. Lee, S. Bratos, M. Wulff, Science 309, 1223 (2005).
Principal publication and authors
Q.Y. Kong (a), M. Wulff (a), J.H. Lee (b), S. Bratos (c), H. Ihee (b), J. Amer. Chem. Soc. 129, 13584 (2007).
(a) ESRF
(b) KAIST, Daejeon (Republic of Korea)
(c) Université Pierre et Marie Curie, Paris (France)



