- Home
- News
- Spotlight on Science
- When carbon meets...
When carbon meets nitrogen under pressure
19-03-2026
Single-crystal X-ray diffraction at beamlines ID15B and ID27 was used to determine the structures of novel nitridocarbonates at extreme conditions. High pressure enabled the formation of previously inaccessible nitrogen-rich carbon frameworks, expanding the chemistry of carbon and nitrogen and providing insight into processes that may occur in planetary interiors.
Share
The challenge
Carbon and nitrogen are among the most versatile elements in the periodic table. Together they form the backbone of life, enable modern catalysts, and are key components of a wide range of functional materials. Their bonding flexibility gives rise to an extraordinary range of molecular and extended structures, from simple small molecules to amino acids and complex framework solids. Under ambient conditions, both elements often adopt exceptionally stable forms, such as graphite, diamond, or molecular nitrogen (N2), which makes their chemical activation and mutual reactivity inherently difficult.
A long-standing question is whether high pressure can overcome these limitations by altering the chemical reactivity of the elements. In particular, can pressure drive carbon and nitrogen to react in the presence of electropositive elements, stabilising chemically complex compounds that cannot be obtained under conventional conditions? Addressing this challenge is essential not only for expanding the boundaries of synthetic chemistry, but also for understanding how unusual compounds may form in planetary interiors and other extreme environments.
The experiment
In a series of experiments performed at beamlines ID15B and ID27, reaction mixtures containing carbon, nitrogen, and selected electropositive elements were compressed to pressures of 30 – 50 GPa in diamond anvil cells, and chemical reactions were induced by laser heating. Investigating the reaction products formed under such conditions requires extremely brilliant and focused X-ray beams, which enable the collection of diffraction patterns from dozens of single crystals produced during the synthesis. This approach allows crystal structures to be solved and refined without prior knowledge of the exact chemical composition, space group symmetry, or structural building blocks.
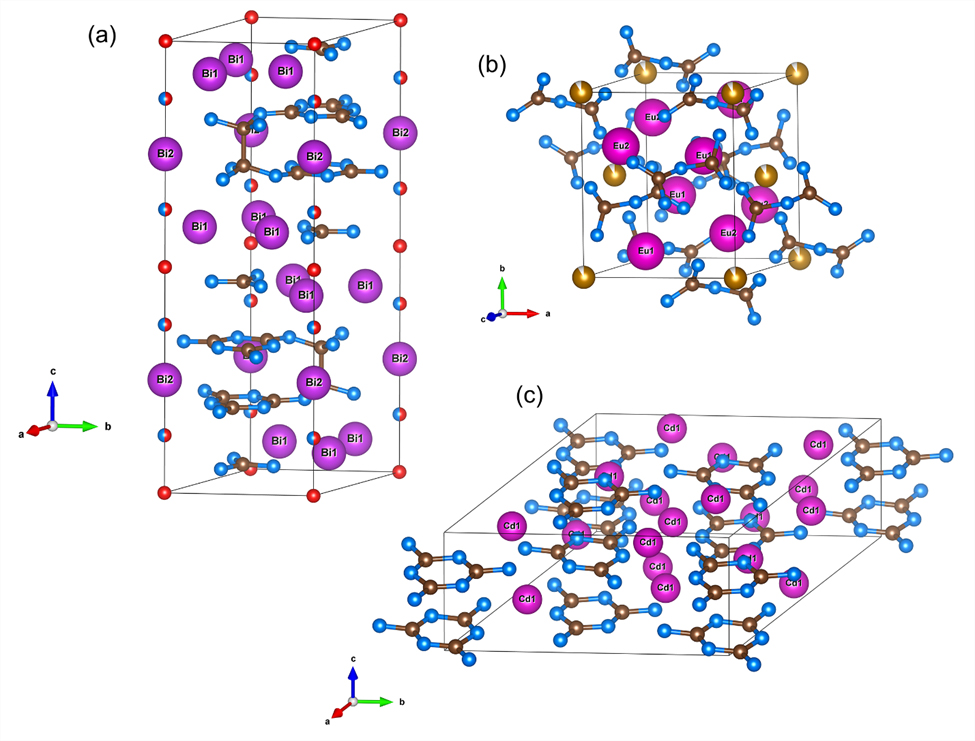
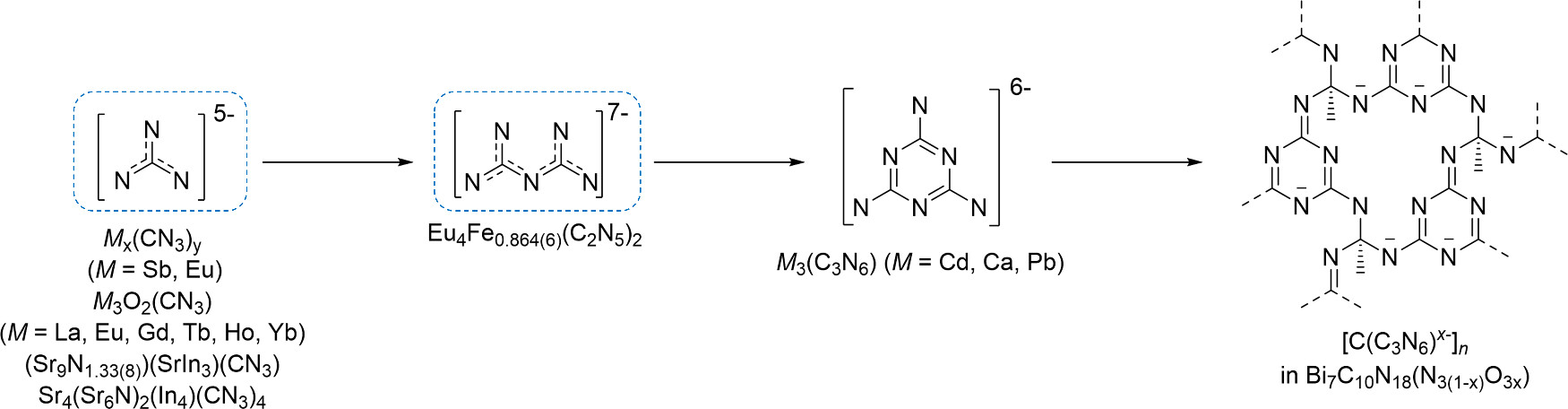
Unprecedented nitridocarbonates were produced from a variety of precursors, including simple mechanical mixtures of elements (Cd + C + N2), single-source precursors (CaCN2), or more complex starting materials (Bi + C6N4 or Eu(N3)2 + EuC2 + Fe). Building on the simplest guanidinate anions first identified in 2023 [1,2], significantly more complex species are now accessible, including pyronitridocarbonates (Eu4Fex(C2N5)2), melaminates (Cd3C3N6 and Ca3C3N6), and two-dimensional networks such as Bi7C10N18(N3(1-x)O3x) (Figures 1 and 2).
Click image to enlarge
Fig. 1: Crystal structures of (a) Bi7C10N18(N3(1-x)O3x), (b) Eu4Fex(C2N5)2, and (c) Cd3C3N6.
Highly charged anions such as pyronitridocarbonates [C2N5]7- or [C3N6]6- could not be obtained under ambient conditions from their organic parent compounds (biguanide or melamine) through simple deprotonation. Likewise, the two-dimensional C-N networks in Bi7C10N18(N3(1-x)O3x) have no known low-pressure analogues. The formation of these compounds under high-pressure conditions highlights the unique role of pressure in stabilising highly charged, nitrogen-rich anionic species that are inaccessible via conventional synthetic routes.
Click image to enlarge
Fig. 2: Schematic representation of the family of nitridocarbonates featuring progressive oligomerisation of CN3 building blocks: guanidinate [CN3]5–, pyronitridocarbonate [C2N5]7–, melaminate [C3N6]6–, and poly-N-(1,3,5-triazin-2-yl)-guanidine anion [C4N6x–]n.
The impact
The significance of this discovery lies in demonstrating that entirely new structural principles can remain hidden until the right conditions are applied – even for intensively studied elements such as carbon and nitrogen. The pressures used in the experiments are not exotic in a planetary context – they exist naturally deep inside the Earth and other planets. Simple reactions between carbon- and nitrogen-bearing species can generate surprisingly intricate molecules, and such processes may represent important steps on the path from basic elements to the complex chemistry required for life. Furthermore, complex carbon–nitrogen building blocks play a central role in many functional materials. Learning how to create and control them could therefore stimulate future advances in catalysis, electronic materials, and energy technologies.
|
Principal publications [2] Stabilization of Fully Deprotonated Melaminate Anions (C3N6)6− in M3(C3N6) (M = Cd, Ca), P.L. Jurzick et al., J. Am. Chem. Soc. 148, 2843−2850 (2026); https://doi.org/10.1021/jacs.5c16752 [3] Stabilization of the [C2N5]7– Anion in Recoverable High-Pressure Eu4Fe0.864(6)(C2N5)2 Pyronitridocarbonate, F.I. Akbar et al., J. Am. Chem. Soc. (2026); https://doi.org/10.1021/jacs.5c21756
|
|
About the beamlines: Beamline ID15B is dedicated to studying the structural properties of solids under high pressure using angle-dispersive X-ray diffraction with diamond anvil cells. It operates at a photon energy of 30 keV for high-pressure experiments, delivering a flux of 1012 photons/s at 200 mA. The typical beam size at the sample position can be adjusted from collimated to focused, down to 5 × 5 µm2, and can be further reduced to 1 × 1 µm2 for experiments at megabar pressures. The station is equipped with a variety of sample environments, including multiple membrane-driven diamond anvil cells (0 – 100 GPa), a liquid helium-cooled cryostat enabling high-pressure experiments at low temperatures (down to 4 K), and external resistive heating equipment for temperatures up to 1200 K. Additionally, an external Nd:YAG laser system is available for high-temperature annealing of samples inside the diamond anvil cell.
|