- Home
- News
- Spotlight on Science
- How membranes shape...
How membranes shape crystal growth in marine algae
07-04-2026
Cryo-electron microscopy and nano-XRF tomography at beamline ID16A have resolved how coccoliths – calcite plates produced by single-celled marine algae – form within membrane-bound vesicles. Distinct membrane–crystal proximity bands define chemically differentiated compartments that regulate crystal growth, providing new insight into controlled biomineral formation and bio-inspired materials design.
Share
The challenge
Biomineralisation is the process by which organisms produce hardened materials such as bone or shell, a capability that has existed since the earliest life on Earth. Although biominerals share the same basic constituents as geogenic rocks and minerals (e.g., carbonate salts or silicates), they form under strict biological control: in specialised environments, in the presence of organic molecules, and at near-ambient conditions. This enables reproducible growth of composite materials with complex architectures and enhanced physical properties, spanning nanometre to metre scales. Understanding these processes may help guide the design of more sophisticated and sustainable materials.
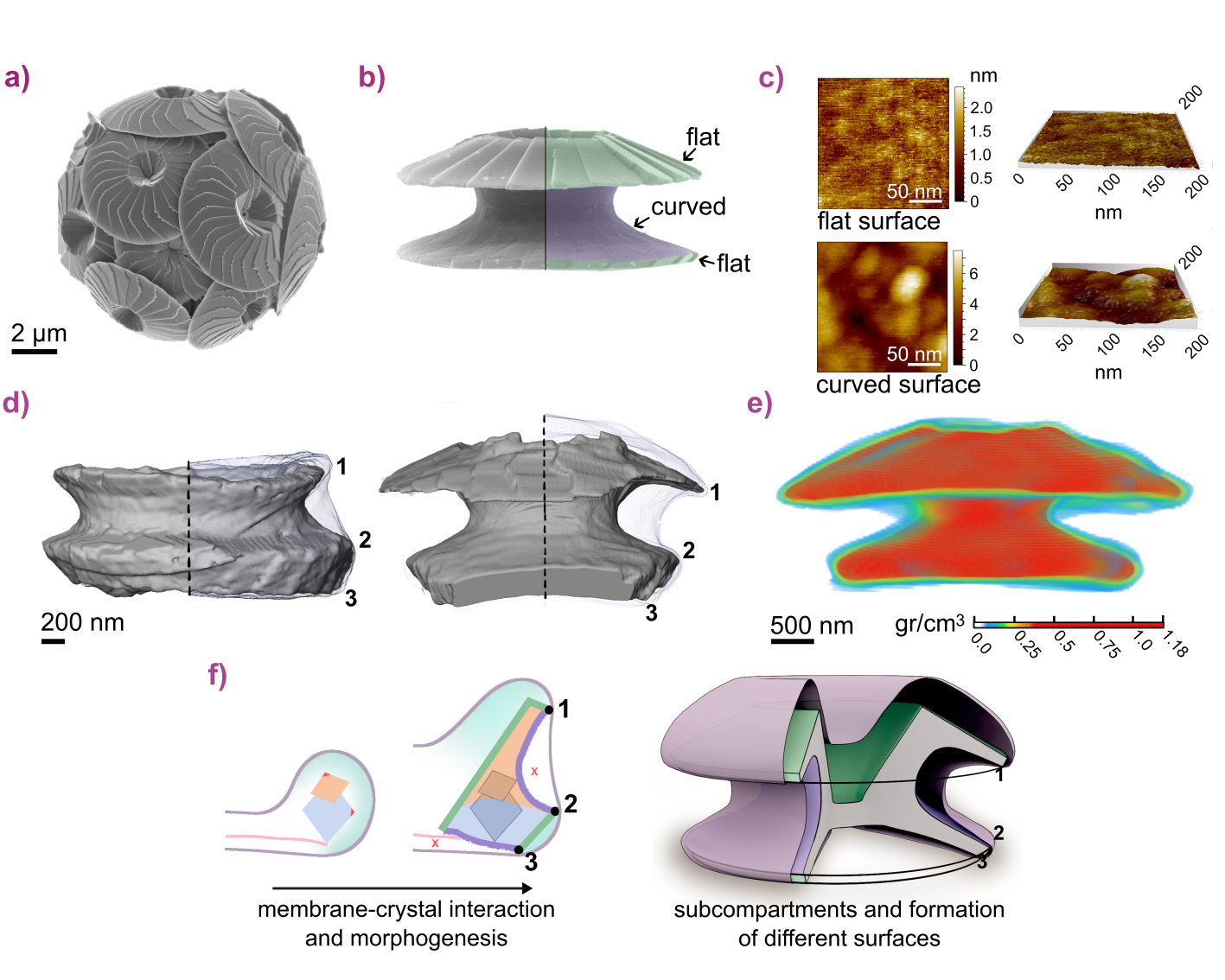
Coccolithophores are ubiquitous unicellular marine algae that produce calcite (CaCO3) plates called coccoliths – multi-crystal arrays with diverse morphologies and surfaces that can be flat or curved (Figure 1a-b). Each coccolith forms intracellularly within a membrane-bound vesicle, where crystals nucleate and grow into intricate shapes. Crystal asymmetry has been attributed to the anisotropic growth environment within this vesicle [1], with distinct crystal surfaces reflecting local growth regimes [2].
However, the spatial relationship between the growing crystals and the surrounding vesicle membrane – which defines the environment and governs morphogenesis – has remained poorly understood. Addressing this requires correlative methods capable of resolving both structural and chemical information at the nanoscale.
The experiment
The morphological evolution of Calcidiscus leptoporus coccoliths was characterised both ex situ and in situ within the coccolith vesicle of cryo-fixed cells preserved in a near-native-state. This species produces relatively large coccoliths with both flat and curved surfaces, making it well-suited to probing spatial relations between crystals and membrane.
Atomic force microscopy (AFM) measurements on extracted coccoliths revealed clear topographical differences between flat (crystallographic) and curved surfaces (smooth and rough, respectively), indicating distinct growth environments (Figure 1c). In situ cryogenic focused ion beam scanning electron microscopy (FIB-SEM) imaging of whole vesicles, combined with high-resolution cryogenic transmission electron microscopy (TEM) tomography of selected lamellae, enabled three-dimensional reconstruction of both crystals and membrane (Figure 1d).
Click image to enlarge
Fig. 1: a) Calcidiscus leptoporus cell. b) Side view of an individual coccolith. c) AFM scans showing distinct topographies of flat and curved surfaces (corresponding to those in (b)). d) 3D renderings of coccoliths within the vesicle from cryo-FIB-SEM. Points 1-3 represent regions of contiguous high proximity between the membrane and the crystals. e) 3D calcium distribution from reconstructed tomography of a whole coccolith within the vesicle, acquired with cryo-nano-XRF. f) Model of compartmentalised growth defined by membrane–crystal proximity bands.
These data show that the membrane adopts distinct spatial patterns relative to the evolving crystals. Regular zones of close membrane–crystal proximity coincide with transitions between surface types (Figure 1d, points 1-3), while regions of variable separation correspond to areas where different surfaces develop. This spatial organisation indicates that crystal growth is locally regulated within sub-compartments of a single vesicle.
To test whether this structural compartmentalisation reflects chemically distinct environments, nanoscale elemental sensitivity was required. Cryogenic nano-X-ray fluorescence (XRF) computed tomography at beamline ID16A – enabled by the EBS and advanced correlative cryogenic workflows [3] – provided this capability.
Whole-cell XRF tomograms acquired at 30 nm voxel resolution captured the full volume of coccoliths within their vesicles. Combined with electron microscopy, these data revealed inhomogeneous calcium distributions across the vesicle interior, spatially correlated with membrane-crystal proximity bands, confirming the presence of chemically distinct growth domains (Figure 1e).
The impact
These findings demonstrate that coccolith formation proceeds through sub-compartmentalisation within a single vesicle, mediated by contiguous membrane proximity bands along the multi-crystal structure. These nanoscale domains create distinct growth environments that regulate crystal morphology (Figure 1f).
This mechanism suggests a general strategy by which biological systems constrain degrees of freedom during biomineralisation, increasing robustness to perturbations. More broadly, these results enhance our understanding of how complex biogenic crystals form under near-ambient conditions and provide a framework for future bio-inspired synthesis.
Principal publication
Spatial Relations between Coccoliths and Their Confining Membrane During Crystal Morphogenesis, E.M. Avrahami et al., J. Am. Chem. Soc. (2026); https://pubs.acs.org/doi/10.1021/jacs.6c02151
References
[1] E.M. Avrahami et al., Science 376, 312-316 (2022).
[2] E.M. Avrahami et al., Adv. Mater. 36, 2309547 (2024).
[3] D. Karpov et al., ACS Nano 20, 7401-7413 (2026).
|
About the beamline: ID16A The beamline provides a high-brilliance beam focused down to a few tens of nanometres to perform coherent imaging techniques, X-ray fluorescence microscopy, and nanotomography. All measurements can be performed under cryogenic conditions to preserve the biological samples close to their native hydrated state and reduce radiation damage. |