- Home
- News
- Spotlight on Science
- How manganese disrupts...
How manganese disrupts neuronal function
12-05-2026
Excess manganese can be neurotoxic, but intracellular distribution within brain cells remains poorly understood. High-resolution X-ray fluorescence imaging at beamline ID21 was used to map manganese inside neurons, revealing preferential accumulation in the Golgi apparatus, partial buffering by astrocytes, and possible effects on synaptic compartments. These findings provide new insights into manganese neurotoxicity and may contribute to improved strategies for limiting exposure and protecting brain health.
Share
The challenge
Manganese (Mn) is an essential element required for normal physiological function, but excessive exposure can lead to neurotoxic effects [1]. The margin between essentiality and toxicity is narrow, and chronic environmental exposure to Mn is associated with neurodevelopmental and neurodegenerative disorders [2]. While neurons are the primary targets of Mn neurotoxicity, astrocytes may play a dual role in Mn regulation, contributing to both protection and toxicity [3].
A key question was how Mn distributes at the subcellular level in neurons and astrocytes, and how this relates to neurotoxicity. Addressing this required quantitative imaging of Mn at high spatial resolution in neurons and astrocytes exposed to sub-cytotoxic doses of Mn. Synchrotron X-ray fluorescence imaging using the nano-scale scanning X-ray microscope at beamline ID21 provided the necessary analytical sensitivity and spatial resolution to map elemental distributions within subcellular compartments of individual cells.
The experiment
Synchrotron X-ray fluorescence imaging at ID21 was combined with cryo-fluorescence light microscopy to map Mn distribution in primary rat hippocampal neurons and astrocytes exposed to sub-cytotoxic Mn doses. Two experimental conditions were compared: neurons cultured alone and neurons co-cultured with astrocytes. Fluorescence labelling identified three subcellular compartments: the nucleus, the Golgi apparatus, and the postsynaptic density (Figure 1a-b).
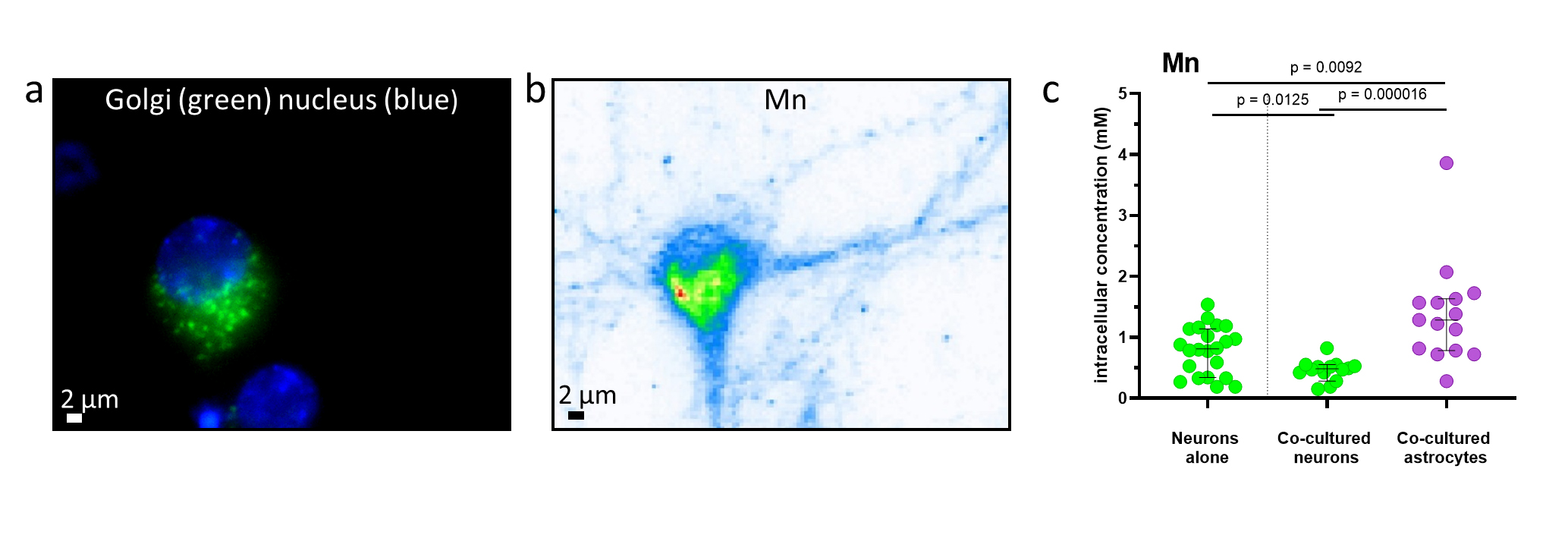
Fig. 1: Manganese imaging and quantification. a) Optical fluorescence image showing the nucleus (blue) and Golgi apparatus (green) in a primary neuron. b) Corresponding Mn distribution in the same cell measured by synchrotron X-ray fluorescence. c) Quantification of Mn in single cells; each point represents one neuron (green) or astrocyte (magenta). Black lines indicate median values and 95% confidence intervals; p-values are shown on top of the figure. Astrocytes accumulate more Mn than neurons and reduce neuronal Mn uptake in co-culture.
The Golgi apparatus was identified as the main site of Mn accumulation in both neurons and astrocytes. Astrocytes accumulated approximately three times more Mn than neurons, and neuronal Mn uptake decreased in co-culture, indicating a buffering and potential neuroprotective role of astrocytes against Mn toxicity (Figure 1c). In neurons, Mn was also detected in postsynaptic compartments, suggesting a potential mechanism of synaptic disruption (Figure 2).
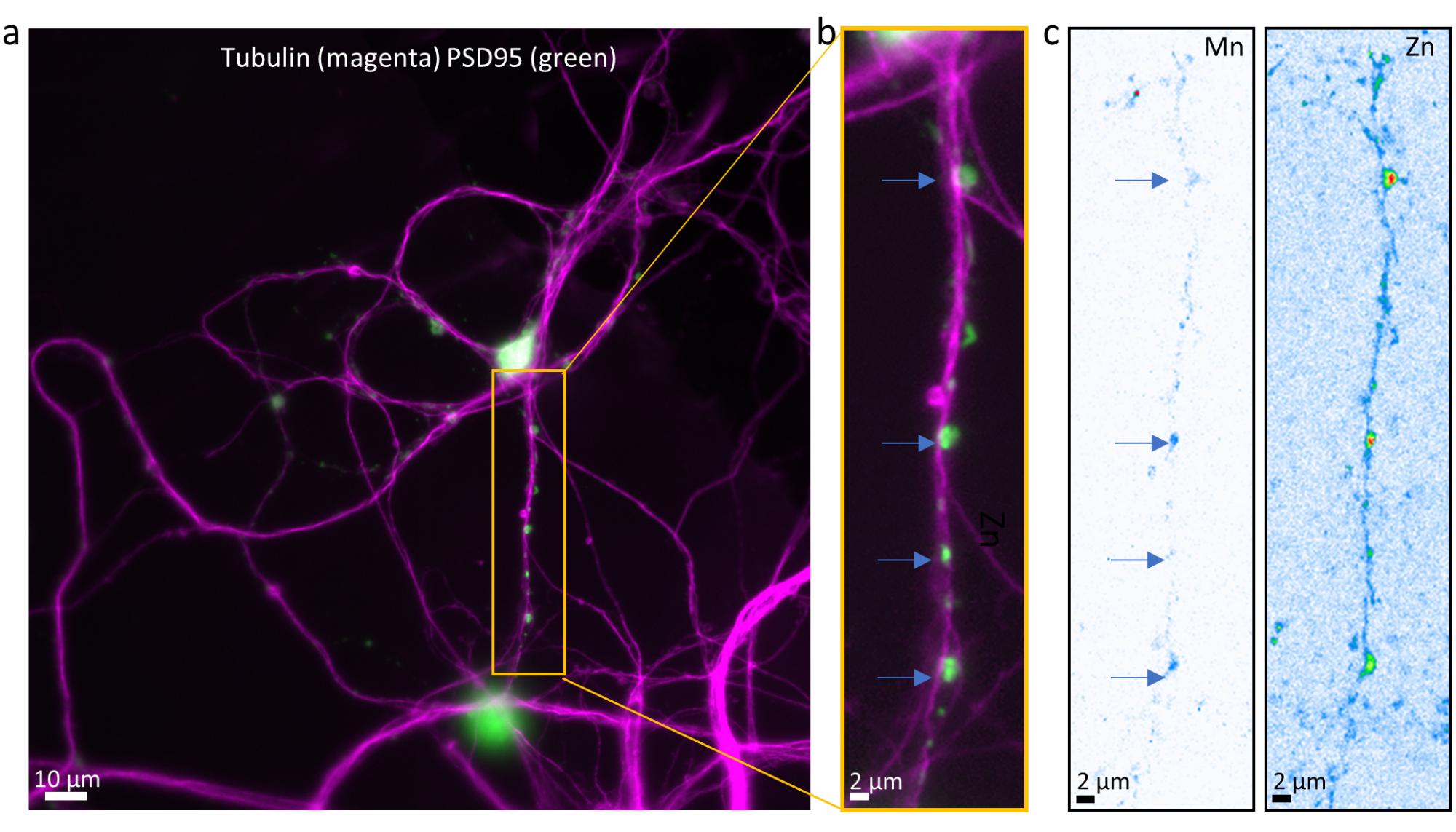
Fig. 2: Detection of manganese in synaptic compartments by nano-synchrotron X-ray fluorescence.
a-b) Optical fluorescence images of tubulin (magenta) and PSD-95 (green) in primary hippocampal neurons. c) Elemental maps of Mn and Zn in the same region as panel (b). Arrows indicate Mn localisation within postsynaptic compartments.
The impact
This work identifies the Golgi apparatus as a major site of intracellular manganese accumulation, providing a possible explanation for toxicity through disruption of protein processing or vesicular transport. The observed buffering capacity of astrocytes highlights their protective role in limiting neuronal exposure and suggests potential detoxification pathways. The presence of Mn in synaptic compartments indicates that synaptic connectivity may be directly impaired, likely contributing to neurotoxicity. Overall, these findings advance the understanding of manganese toxicity mechanisms and provide a basis for future studies on mitigation strategies.
|
Principal publication References |
About the beamline: ID21
ID21 is dedicated to micro- and nano-X-ray spectroscopy, offering 2D X-ray fluorescence (XRF) mapping and X-ray absorption spectroscopy (XAS) in the tender X-ray energy range (2.1 - 10.5 keV). These techniques can be combined to produce multi-energy XRF maps, enabling both 2D elemental mapping and chemical speciation across point (0D), line (1D), and area (2D) measurements. The beamline is optimised for detecting and chemically analysing elements from phosphorus to zinc; heavier elements can also be studied via their L- and M-edges.
Following a recent upgrade, ID21 now hosts two scanning X-ray microscopes. A newly installed nanoscope (nano-SXM) is optimised for high-resolution (down to 120 nm), high-speed nano-XRF mapping and nano-XAS, offering enhanced performance over the previous microscope. The original microscope has been retained and is now dedicated to larger samples, using micrometric and submillimetric beams. Both microscopes operate under vacuum and support room temperature and cryogenic measurements.