- Home
- News
- Spotlight on Science
- High-temperature...
High-temperature molecular magnetism unlocked with X-ray dichroism
08-12-2025
Molecule-based magnets offer tuneable optical and electronic properties for multifunctional materials, but achieving magnetic ordering above room temperature is challenging. X-ray magnetic circular dichroism at ID12 revealed the antiparallel alignment of vanadium and molybdenum magnetic moments in a newly synthesised Prussian Blue analogue, establishing the system as a platform for robust, high-temperature molecular magnets.
Share
The challenge
Molecule-based magnets are promising candidates for future information technologies because they combine magnetic, optical [1], and electronic functionalities within a single, chemically tuneable material [2]. Prussian Blue analogues (PBAs), composed of metal ions bridged by cyanide ligands, are a versatile testbed for exploring multifunctional molecular systems. However, most molecular magnets order only at cryogenic temperatures, far below the threshold for practical applications, with very few known exceptions [3]. Achieving magnetic ordering above ambient temperature has long remained a key scientific challenge.
For nearly twenty years, theory has suggested that substituting chromium (CrIII) with the heavier molybdenum (MoIII) ion in a cyanide-bridged vanadium-chromium Prussian Blue analogue, hexacyanomolybdate (VII-CrIII(CN)6 –> VII-MoIII(CN)6) PBA could dramatically increase the magnetic ordering temperature [4]. Yet the idea remained untested because MoIII cyanometallates decompose rapidly in protic solvents – including water, the standard medium for PBA synthesis. As a result, no experimental proof of this long-standing prediction was available. This study finally resolves this open question by using element-specific X-ray magnetic circular dichroism (XMCD) spectroscopy to directly probe the alignment of the magnetic moments of metal ions in a newly synthesised VII-MoIII(CN)6 PBA.
The experiment
First, the VII-MoIII(CN)6 PBA was synthesised, using mechanochemical grinding rather than solution chemistry, thereby bypassing the aqueous conditions that previously caused MoIII decomposition. The resulting amorphous powder exhibited strong magnetism at room temperature.
To determine the nature of the long-range magnetic order, XMCD was employed at beamline ID12. The beamline provides intense, highly circularly polarised X-rays and is one of the few facilities capable of performing XMCD at the Mo L2,3 edges. These measurements provided element-specific magnetic signatures that helped to identify the orientation of vanadium and molybdenum magnetic moments.
Click image to enlarge
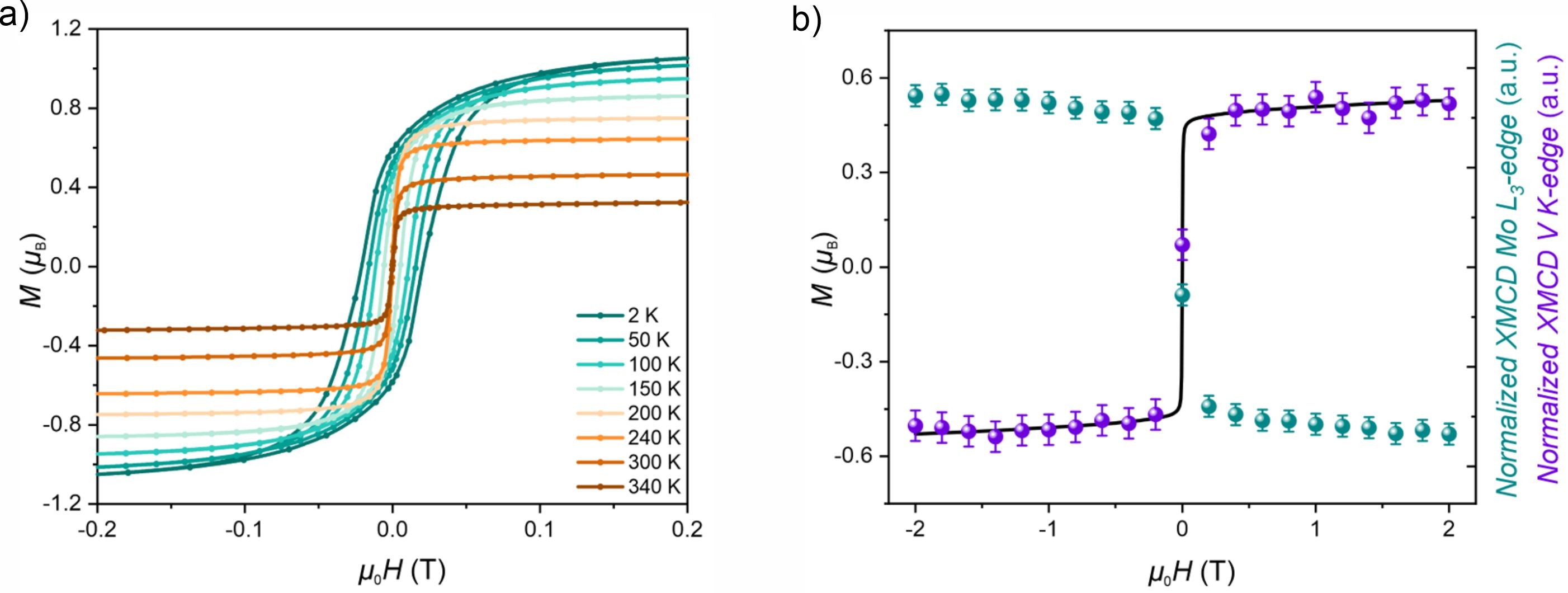
Fig. 1: a) Variable-temperature magnetic hysteresis loops for the VII-MoIII(CN)6 magnet measured using a SQUID magnetometer. b) Normalised XMCD signals at the Mo L3-edge (green) and V K-edge (violet) at room temperature, compared with the magnetic hysteresis loop recorded at 300 K (black), confirming ferrimagnetic ordering.
The XMCD spectra revealed an antiparallel alignment of the V and Mo magnetic moments – an unequivocal fingerprint of ferrimagnetic ordering (Figure 1b). Complementary superconducting quantum interference device (SQUID) magnetometry confirmed a critical temperature exceeding 400 K, far above room temperature and among the highest ever reported for a molecule-based magnet (Figure 1a). Together, the results show that hexacyanomolybdateIII can indeed stabilise high-temperature magnetic order, validating the long-standing theoretical prediction (Figure 2).
Click image to enlarge
Fig. 2: A frame from a demonstration showing a VII-MoIII(CN)6 sample being attracted to a 0.5 T electromagnet (full video available in the Supporting Information: https://doi.org/10.1002/advs.202511285).
The impact
Demonstrating that MoIII-based PBAs can sustain magnetic ordering well above room temperature opens an entirely new chemical landscape for molecule-based magnets. The discovery removes a key obstacle to designing cyanide-based materials that combine magnetic, optical, and electronic functions under technologically relevant conditions.
Such robust, high-temperature molecular magnets could support advances in low-energy data storage, spintronic devices, and multifunctional materials engineered at the molecular level. More broadly, the study illustrates how combining innovative synthesis routes with element-specific X-ray techniques enables breakthroughs that were previously considered inaccessible.
Principal publication
Heavy Prussian Blue Analog with Magnetic Ordering above 400 K, M. Magott et al., Adv. Sci. e11285 (2025); https://doi.org/10.1002/advs.202511285
References
[1] J.J. Zakrzewski et al., Chem. Rev. 124, 5930 (2024).
[2] S. Chorazy et al., Chem. Soc. Rev. 49, 5945 (2020).
[3] S. Ferlay et al., Nature 378, 701 (1995).
[4] E. Ruiz et al., Chem. Eur. J. 11, 2135 (2005).
| About the beamline: ID12 |
|
Beamline ID12 specialises in polarisation-dependent X-ray spectroscopy within the tender and hard X-ray range (2 -15keV), offering full control over the polarisation state of the X-ray beam. Its research activities focus on investigating the electronic and magnetic properties of diverse systems, ranging from bulk permanent magnets and diluted magnetic semiconductors to paramagnetic monolayers on surfaces, with element- and orbital-selective sensitivity. A wide array of dichroic experiments can be performed, probing magnetism, chirality, or both, under extreme conditions, including magnetic fields up to 17 T, temperatures spanning from 2K to 800K, and pressures reaching 60 GPa. The beamline’s exceptional optical stability, combined with a highly efficient detection system, enables the reliable measurement of dichroic signals with an unprecedented signal-to-noise ratio. Using a focused ESRF-EBS beam, twin domain mapping in chiral systems can be conducted with micrometric resolution. Additionally, the ULMAG setup allows users to perform X-ray magnetic circular dichroism, X-ray diffraction, and bulk property measurements – including magnetisation, magnetostriction, magnetocalorics, and magnetoresistance – under strictly identical experimental conditions. This unique and versatile tool provides crucial insights into the interplay between magnetic, structural, and electronic subsystems in a wide range of magnetic materials. |