- Home
- News
- General News
- X-rays reveal the...
X-rays reveal the oxidation state of uranium
19-12-2013
Uranium is often found in several different oxide forms. Understanding how uranium oxides convert from one form to another is important for nuclear waste disposal. A new study of two uranium oxides using high brilliance X-rays has revealed an oxidation state of uranium that had not previously been observed directly. The results should improve models for how uranium behaves in geochemical processes and how it might work as a catalyst for industrial purposes.
Share
Nuclear fuel rods, which are used to derive nuclear energy, typically contain uranium dioxide (UO2). After use, the spent uranium dioxide is often stored in containers. Uranium dioxide will gradually oxidize with time into mixed oxides which have more than one oxidation state and whose behaviour is not well known.
These uranium oxide reactions can lead to environmental contamination so learning how the different oxide compounds behave in the final oxidation states is important for understanding how to dispose of nuclear waste.
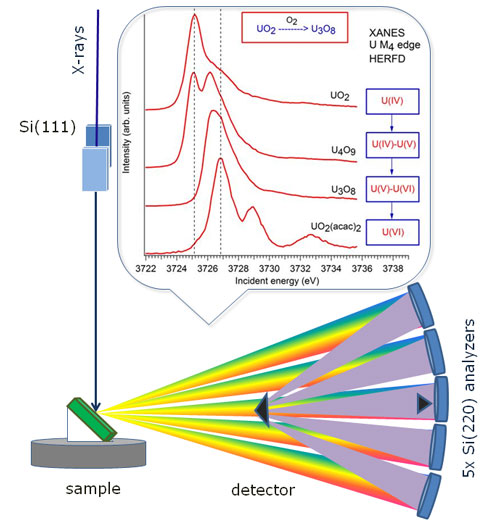
Top: Simplified single electron scheme of the transitions at the U M4 edge for the different oxidation states. Bottom: Schematic drawing of the X-ray emission spectrometer used during the high energy resolution fluorescence detection experiments.
The research team from the European Synchrotron in Grenoble (France), led by Kristina Kvashnina, performed X-ray spectroscopy experiments on the mixed oxides in order to study the oxidation state of the mixed uranium oxides, such as U4O9 and U3O8. They probed the transition from the 3d electron shell to the 5f shell in the uranium atom, which requires exposing samples to low energy 3.7 KeV X-rays.
“The brilliant X-rays unique to the ESRF and high energy resolution X-ray emission spectrometer at ID26 beamline enabled us to identify the element U(V) as one of the oxidation states.” explains Kristina Kvashnina, and adds “This observation calls for a revision of the current thinking on uranium chemistry”.
References
Chemical State of Complex Uranium Oxides, K.O. Kvashnina, S.M. Butorin, P. Martin and P. Glatzel, Phys.Rev. Lett. 111, 253002 (2013), 17 December 2013.
DOI: 10.1103/PhysRevLett.111.253002
Top image: Kristina Kvashnina sets up the experiment on ID26. Credit: Molyneux Associates



