- Home
- News
- General News
- Water and cations...
Water and cations play tag in electrolysers
09-04-2024
Researchers led by the Technical University of Denmark, in collaboration with the ESRF, have unveiled how water and Cessium+ ions move inside a membrane electrode assembly in a zero-gap CO2 electrolyser. They used wide-angle X-ray scattering and X-ray fluorescence at the ESRF. The results are out in the journal Joule.
Share
CO2 electrolysis is a promising approach to produce high-value products from CO2, which is one of the primary contributors to global warming. In particular, zero-gap CO2 electrolysers have the ability to produce desired electrolysis products with high efficiency and performance.
Unlike traditional electrolysers that have a physical gap between the electrodes, zero-gap electrolysers use a membrane electrode assembly (MEA) to separate the electrodes while allowing ions to pass through thin polymer membrane. This design reduces electrical losses through resistance and increases efficiency. However, current CO2 electrolysis efforts using MEA technology show that it has an efficiency of 34% and operating lifetimes of tens to a few hundred hours.
The precise engineering of the chemical balance and mass transfer balance in these electrolysers is key to the design of devices that are more efficient. On the cathode, the core part of the electrolyser, the balance of water and cations is crucial. One of the known challenges of these systems is that excessive accumulation of water and cations causes performance degradation, leading to flooding and salt precipitation. This has a direct impact on performance.
Now scientists led by the Technical University of Denmark (DTU), in association with the ESRF, have observed what takes place inside the membrane electrode assembly (MEA) while the electrolyser is operating.
They came to the ESRF’s ID31 beamline to study zero-gap electrolysers using wide-angle X-ray scattering and X-ray fluorescence. “The ability to observe the movement of cations and water within MEA during operation is truly extraordinary, presenting a unique opportunity to gain valuable insights into our devices”, says Bjørt Joensen, PhD student at DTU and first author of the publication. Jakub Drnec, scientist in charge of ID31, explains how the experiment unfolded: “The main challenge of these experiments is the complexity of the device; therefore we had to use multiple characterization strategies at the same time to properly describe what is actually going on inside the electrolyser during operation”.
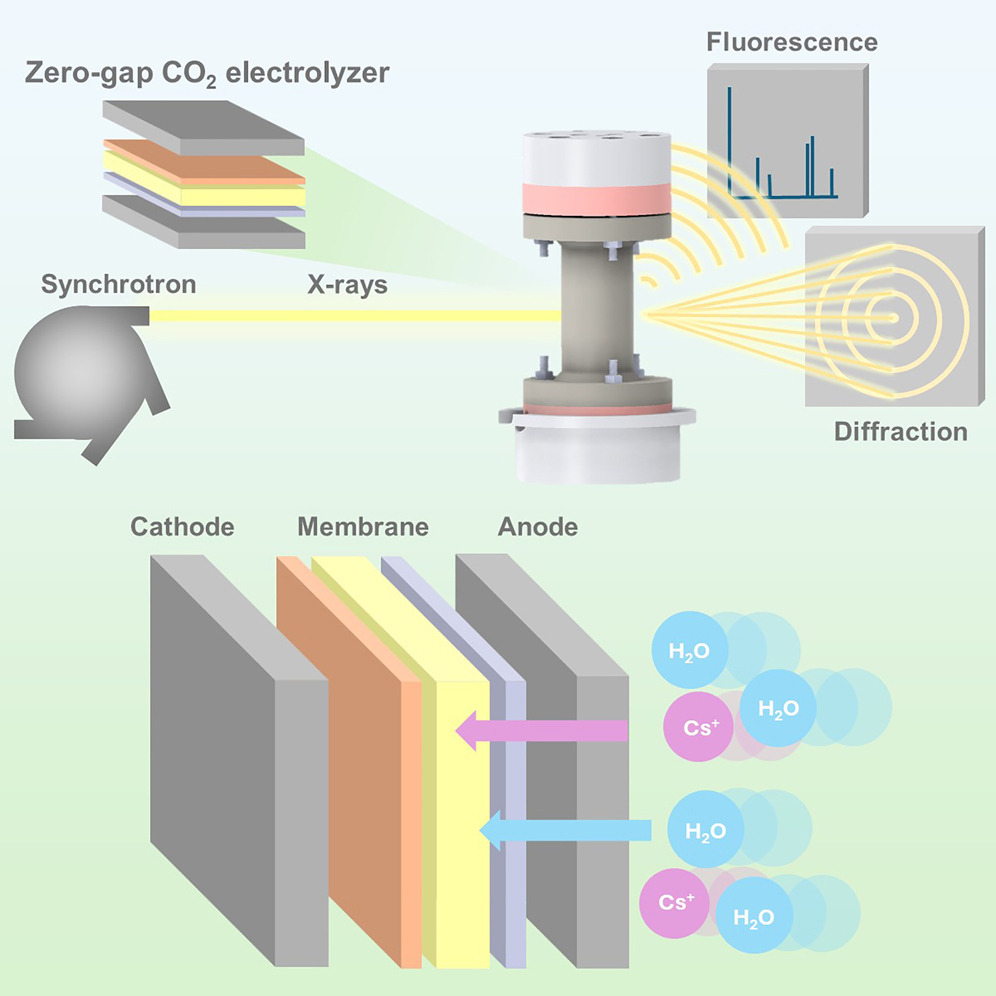 |
|
Graphical abstract of the experiment. Credits: B. O. Joensen et al, Joule, In Press, Corrected Proof, 18 March 2024. |
They found that Cessium+ ions travel from anode to cathode, dragging water with them, in a phenomenon called electroosmosis, as if they were playing the children’s game tag. Once in the gas diffusion electrode (GDE), the water diffuses further. When the scientists adjusted the voltage, Cessium+ rushed back to the anode, whilst water stayed put in the GDE, which lead to flooding.
“There is an intricate balance on water and cations in the CO2 reduction, and now we’ve found how the flooding and the salt precipitation are triggered. With this information we can define new strategies to regulate electrolyte concentration in a certain way if we want the electrolysers to work properly”, explains Qiucheng Xu, post-doctoral researcher and co-corresponding author of the publication. And he adds: “We are sure the continuous cooperation with ID 31 beamline will further enhance our understanding and optimise the CO2 electrolysis system”.
These findings have a direct impact on commercialization of the CO2 electrolysers, as they show the way to operate such devices and may lead to devise strategies for mitigation of salt formation and flooding, significantly increasing the performance and lifetime of the electrolysers.
Reference:
B. O. Joensen et al, Joule, In Press, Corrected Proof, 18 March 2024. https://doi.org/10.1016/j.joule.2024.02.027
Text by Montserrat Capellas Espuny
Top image: The set-up on ID31. Credits: Qiucheng Xu.



