- Home
- News
- General News
- The mechanism of...
The mechanism of the most commonly used antimalarial drugs unveiled in near-native conditions
28-10-2019
For centuries, quinoline has been an effective compound in antimalarial drugs, although no one knew its mode of action in vivo. Today, a team led by the Weizmann Institute has discovered its mechanism in infected red blood cells in near-native conditions, by using the ESRF, Alba Synchrotron and BESSY. They publish their results in PNAS.
Share
Malaria remains one of the biggest killers in low-income countries. Estimates of the number of deaths each year range from 450,000 to 720,000, with the majority of deaths happening in Africa. In the last two decades, the malaria parasite has evolved into drug-resistant strains. “Recently, the increasing geographical spread of the species, as well as resistant strains has concerned the scientific community, and in order to improve antimalarial drugs we need to know how they work precisely”, explains Sergey Kapishnikov, from the University of Copenhagen, in Denmark, and the Weizmann Institute, in Israel, and leader of the study.
Plasmodium parasite, when infecting a human, invades a red blood cell, where it ingests hemoglobin to grow and multiply. Hemoglobin releases then iron-containing heme molecules, which are toxic to the parasite. However, these molecules crystallise into hemozoin, a disposal product formed from the digestion of blood by the parasite that makes the molecules inert. For the parasite to survive, the rate at which the heme molecules are liberated must be slower or the same as the rate of hemozoin crystallization. Otherwise there would be an accumulation of the toxic heme within the parasite.
 |
|
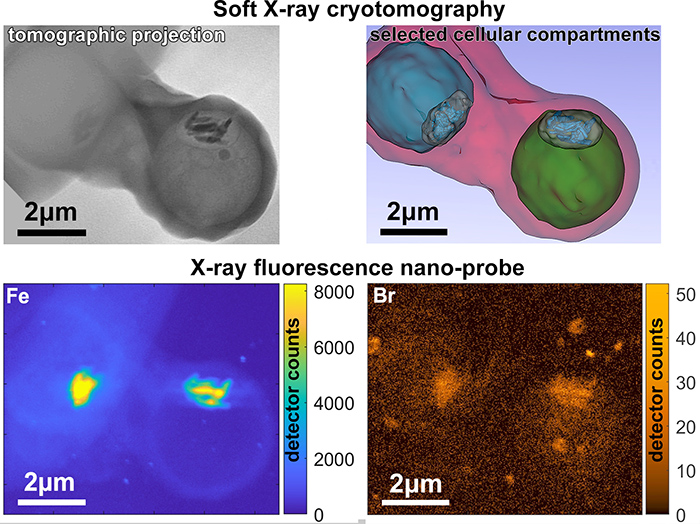
The same infected blood cell (with parasites in blue and green in the top right picture) in correlatively mapped by two methods: SXT at BESSY-II and X-ray fluorescence nano-probe at ESRF. Credits: S. Kapishnikov. |
Quinoline-family drugs, which include quinine-based antimalarial pills, effectively damage the parasite. The scientific community has suspected that the reason for their success is the inability of the heme to crystallise. Until today, all studies of the drug action on heme crystals have been done either on model systems or on dried parasites, which yielded limited data and opened room for speculation. Kapishnikov and his team, which include scientists from Denmark, Spain, Germany, Israel and France, decided to find out the mode of action of established drugs like chloroquine (although they used the analog bromoquine) in fully hydrated, rapidly frozen, Plasmodium falciparum-infected red blood cells.
 |
|
Yang Yang, an ESRF scientist that took part in the study, on beamline ID16A. Credits: S. Candé. |
Rapid freezing creates instant snapshots of the living stage of the cells such that chemical distribution therein is unaltered by sample preparation. This is definitely an advantage but also a challenge because, once frozen, the cells must be maintained under cryogenic conditions at all times. In this case, the same cells had to travel across Europe. The researchers brought them in and out of synchrotron facilities, in order for their structure to be mapped in three dimensions by soft X-ray cryo-tomography at BESSY-II and ALBA, their coordinates on sample grids recorded, and, finally, brought to ESRF for mapping of bromine and iron distribution by the X-ray fluorescence nano-probe on ID16A. The synchrotron data were analysed back in Denmark where scientists determined the correlation between the different imaging modalities and they calculated and interpreted the concentrations of bromoquine at the surface of hemozoin crystals, at the membrane and within the lumen of the parasitic digestive vacuole – the site of the drug action.
|
|
|
The picture Laveran is a 1880 drawing of the malaria parasites by Charles Alphonse Laveran, a French doctor and a scientist, who studied blood of a soldier who passed away from malaria. Laveran observed the soldier's blood in a crude miscroscope at a field hospital and was probably the first scientist to recognise and draw the malaria parasites. |
The mapped infected red blood cells at the synchrotrons showed that that bromoquine caps hemozoin crystals, thereby inhibiting the hemozoin crystal growth and hence, sabotaging heme detoxification. Surprisingly, they also found that bromoquine accumulates in the digestive system of the parasite, which enhances the drug’s efficiency in depriving heme from docking onto the hemozoin crystal surface.
“These results show a model that can be generalized to all quinoline drugs, such as quinine, and our approach can be extended to other families of antimalarial drugs, such as artemisinins”, explains Kapishnikov. “We hope that this knowledge will let us go a step further in designing new, effective drugs against resistant malaria strains”, he concludes.
Reference:
Kapishnikov, S. et al, PNAS, xxxx, www.pnas.org/cgi/doi/10.1073/pnas.1910123116.
Text by Montserrat Capellas Espuny
Top image: A malaria-infected red blood cell bursting. Credits: istockphoto