- Home
- News
- General News
- Newly discovered...
Newly discovered form of salty ices may exist on icy moons
20-02-2023
An international collaboration lead by University of Washington and ESRF, has found two new kinds of hydrates in icy satellites by reproducing the conditions of temperature and pressure of the oceans in the moons. They have used two synchrotron sources, including the ESRF, and their results are published today in PNAS.
Share
Could there be life beyond Earth? Could moons host habitable environment and how did they evolve over time? Can we decipher the history of the solar system? What chemical processes take place in these icy bodies?
Icy satellites are moons that have a surface primarily composed of water ice, or a mixture of water ice and other substances such as salts and organic compounds. These satellites are in the outer solar system, beyond the orbit of Mars, and are thought to have formed from the accretion of icy particles and gas around their host planets. Some examples include Europa and Ganymede, two of Jupiter's moons, and Enceladus and Titan, two of Saturn's moons.
The space missions Galileo, first, and Cassini-Huygens later, have greatly expanded our understanding of icy satellites. In particular, they confirmed the presence of liquid water-based oceans inside these bodies, located underneath a crust of ice.
“These are the only planetary bodies other than the Earth where liquid water is stable at geological timescales, which is crucial for the emergence and development of life. They are, in my opinion, the best place in our solar system to discover extraterrestrial life, so we need to study their exotic oceans and interiors to better understand how they formed, evolved and can retain liquid water in cold regions of the solar system, so far away from the Sun”, explains Baptiste Journaux, professor at the University of Washington and first author of the publication.
Synchrotrons to study extraterrestrial water
Presumably, the water in these oceans contains table salt (sodium chloride), which acts as a natural antifreeze, lowering the freezing point of water and allowing it to remain liquid at temperatures that would normally freeze pure water. However, planetary scientists had, until now, not been able to clearly identify it at the surface, despite using NASA space mission’s surface reflectance infrared spectroscopy. None of the compounds known would match the icy moon surface spectra.
Journaux explains the challenges: “Salt and water are very well known in common conditions. However, beyond that, we are totally in the dark. Now we have these planetary objects that probably have compounds that are very familiar to us, but at very exotic conditions. We have had to redo all the fundamental mineralogical science done in the 19th and early 20th century but at high pressure and low temperature”.
Due to these conditions, the water, which is thought to have a rich composition, crystallises, forming pure water ice and hydrates. When crystallised, it either sinks down to the mantle or goes up to the crust.
The international collaboration of scientists from the University of Washington, the ESRF, DESY, the Institute of Geochemistry and Petrology in Switzerland, the University of Bayreuth in Germany, NASA’s Jet Propulsion Laboratory, and the University of Chicago came to the ESRF to study these solid samples using X-ray diffraction.
“Our goal was to understand which compounds form in the high pressure and low temperature conditions of the icy moons. In our experiments, we used single crystal X-ray diffraction to understand how the new hydrates are organized on atomic scale. It is a superior and unique technique to get unambiguous information on a solid’s crystal structure”, says Anna Pakhomova, scientist at the ESRF and author of the publication.
The researchers recreated the conditions on high-pressure beamline ID15B by using diamond anvil cells and placing them into the cryostat, where they squeezed the liquid up to 25000 times the standard atmospheric pressure. The samples, which consisted of table salt dissolved in water, soon crystallised into previously unknown forms of salt hydrates.
New watery compounds
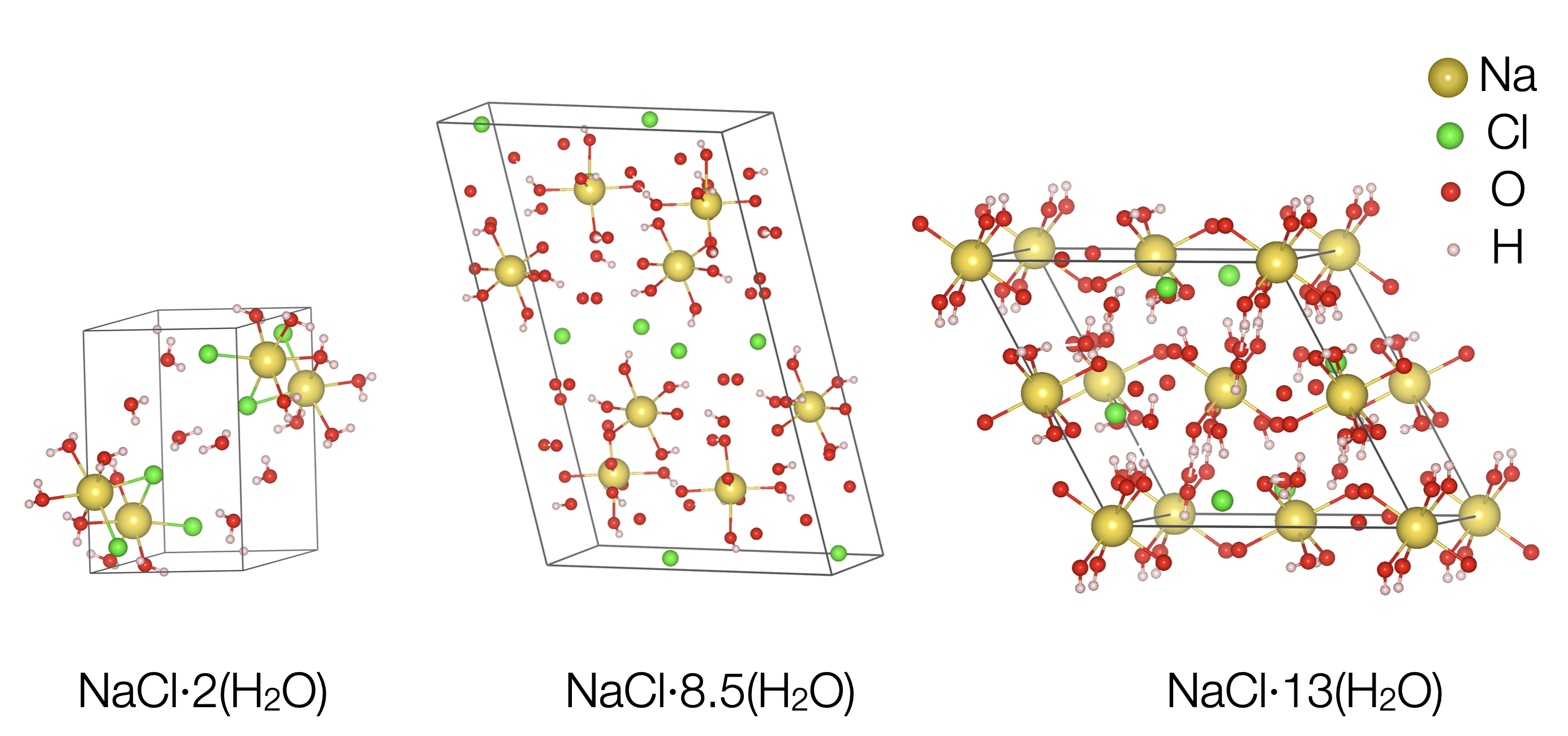
Until then, the scientific community had knowledge about the presence of just one hydrate of sodium chloride, which is a compound that has very few water molecules trapped within its crystal structure. Its composition is a simple structure with one salt molecule for every two water molecules.
The new experiments unveiled two new different stable solid phases, both with a much higher amount of water in the structure and stable at different pressures and temperature. One has two sodium chlorides for every 17 water molecules; the other has one sodium chloride for every 13 water molecules. This would explain why the signatures from the surface of Jupiter’s moons are more “watery” than expected.
“These new phases are fascinating because they demonstrate a new diversity of water/salts crystal structures at high pressure and low temperature that wasn’t expected and remain to be explored for other compounds”, says Journaux.
 |
|
Researchers discovered two new crystals made from water and table salt at low temperatures, below about minus 50°C. The known structure (left) has one salt molecule (yellow and green balls) to two water molecules (red and pink balls). X-ray imaging let researchers determine the position of individual atoms in the new structures. The center structure has two sodium chloride molecules for every 17 water molecules and stays stable even if pressure drops to near vacuum, as would exist on a lunar surface. The structure on the right has one sodium chloride molecule for every 13 water molecules, and is stable only at high pressure. Credit: Baptiste Journaux/University of Washington |
The team was also able to show that one of the new salt hydrates, NaCl·8.5(H2O), is stable at the surface conditions of icy moons, and it should be the most common type of salt hydrate on icy worlds. “It has the structure that planetary scientists have been waiting for to explain the mysterious surface spectra of icy surfaces. This will permit us to identify where are the best places on their surface to explore, and eventually land and dig to look for signs of life”, explains Journaux.
The next step in this research is to study other salt species relevant for icy ocean worlds and measure their spectral properties. This will ensure that they can be detected in upcoming space missions, such as the European Space Agency’s JUpiter ICy moon Explorer (JUICE), launching this April and NASA’s Europa clipper, in 2024, and both entering the Jupiter orbit in the early 2030s.
Journaux, B. et al, PNAS, 21 February 2023. https://doi.org/10.1073/pnas.2217125120
Text and video by Montserrat Capellas Espuny.
Copyright images for video: ESA, NASA.
.
Top image: Jupiter's ocean moon Europa, as seen by NASA's Galileo probe. Credits: NASA/JPL-Caltech/SETI Institute.



