- Home
- News
- General News
- How dolphins could...
How dolphins could potentially lead to new antibiotics
17-05-2018
The world is currently living through a multidrug resistance problem, where antibiotics that traditionally work are not effective anymore. A European team of scientists at the University of Hamburg (Germany), University of Munich (Germany), University of Bordeaux (France), University of Trieste (Italy) and University of London (UK) have studied how some peptides in dolphins target bacterial ribosomes and hence, could provide clues about potential new antibiotics.
Share
Proline-rich antimicrobial peptides (PrAMPs) are antibacterial components of the immune systems of animals such as honey bees, cows and, as this study proves, bottlenose dolphins. These peptides are a first response for the killing of bacteria. In humans, antimicrobial peptides (AMPs) mainly kill bacteria by disrupting the bacterial cell membrane, but so far no evidence of PrAMPs has been found. PrAMPs have a different mechanism of action to AMPs: they pass through the membrane of the cell without perturbing it and bind to ribosomes to inhibit protein synthesis.
The European team have been studying the mechanism of action of bacteria killing peptides in animals: “We want to compare PrAMPs from different organisms to mechanistically understand how these peptides inhibit bacteria”, Daniel Wilson explains.
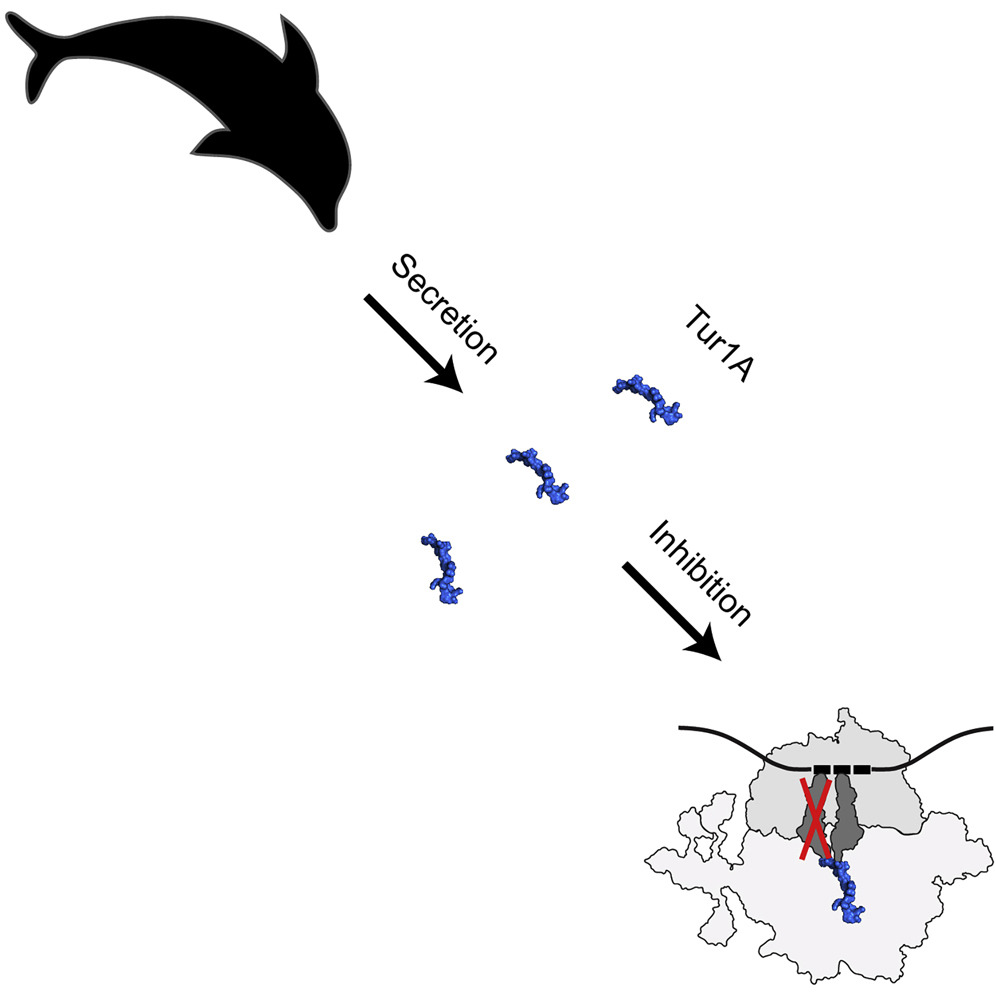
After studying different peptides in cows, among other animals, the team rationalized that similar PrAMPs may also be present in dolphins, since they are co-phyletic with cows within the unique order of Cetartiodactyla. The team discovered two PrAMPs, termed Tur1A and Tur1B, in the bottenose dolphin (Tursiops truncatus). Tur1A was shown to inhibit bacterial protein synthesis by binding to the ribosome and blocking the transition from the initiation to the elongation phase. Despite being a PrAMP, Tur1B was a poor inhibitor of protein synthesis, which could imply that it uses a different as yet unknown mechanism of action.
 |
|
Illustration showing the mechanism of Tur1A. Credits: D. Wilson |
The team came to the ESRF’s ID23-2 beamline to solve the structure of dolphin PrAMPs in complex with the ribosome. “X-ray crystallography was the right technique for us, and the experiment at the ESRF worked wonderfully”, says Natacha Pérébaskine, a member of Axel Innis' team (University of Bordeaux (France).
The results of their research take the scientific community a step closer to the development of new antibiotics: “Both Tur peptides, but especially Tur1A, have a potent antibacterial activity which could be exploited in the fight against the increasing prevalence of antibiotic-resistant pathogens”, says Wilson. “We are currently still studying the mechanisms of PrAMPs in dolphins and other species. Our new findings will definitely contribute to our knowledge of these peptides and will hopefully lead to the design of new antibiotics in the future".
Reference:
Mardirossian, M. et al, Cell Chemical Biology, 17 May 2018. DOI: https://doi.org/10.1016/j.chembiol.2018.02.004
Text by Montserrat Capellas Espuny



