- Home
- News
- General News
- How do I smell?...
How do I smell? Much the same as how you see!
12-09-2014
A collaboration between researchers from the Institute for Medical Physics and Biophysics (IMPB) at the Charité in Berlin and the European Synchrotron (ESRF) has uncovered a new common feature in the molecular processes that are responsible for senses such as taste, smell, and vision. The team discovered that what seem like structurally and functionally different proteins important in sight and smell in fact share a common component. This result, published in the journal Nature Communications, provides another clue in the search to understand how our senses work.
In mammals, the process of ‘sensing’ something is defined by a complex interplay between stimulus, proteins and receptors. For example, in order to see something, the protein rhodopsin must be present – it is responsible for the initial light detection in vision. Rhodopsin belongs to a large protein family called G-protein coupled receptors (GPCRs) which are found in the membranes around every living cell. Their role is to sense, e.g., the presence of molecules outside the membrane, or light, and to amplify and pass on the signal. When light shines on a rhodopsin molecule, the molecule changes shape, creating a new signal pathway. Vision is effectively regulated by the switching on and off of these signal pathways which eventually results in an image being produced in the brain.
In this process, switching-off the signal is an essential step, which happens when the protein arrestin binds to (previously activated) rhodopsin. Interestingly, this latest study from the IMPB Berlin team shows that several different variants of arrestins share a common sequence motif which binds to the GPCR. This suggests very similar interactions between GPCRs and the interaction partners involved in different senses (vision, etc.).
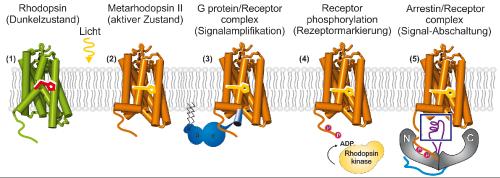
The schema shows the rhodopsin signal transduction: (1) dark state of rhodopsin, (2) after a light signal is received, the activated metarhodopsin-II state is formed, (3) G protein binds to the activated receptor, (4) receptor is phosphorylated, (5) arrestin binds to the phosphorylated receptor. Credit: NPG/authors.
Dr. Patrick Scheerer, Principal Investigator at the Charité in Berlin, and his team determined the X-ray structure of rhodopsin interacting with an arrestin-like molecule. This 3D structure was obtained at the ESRF in Grenoble from tiny protein crystals; a task which wouldn’t have been possible without the use of bright X-ray sources. These results were confirmed using complementary spectroscopic methods, which allowed ESRF researcher Dr. David von Stetten to observe structural changes of the protein under natural conditions. Speaking about the team’s conclusions, lead author Michal Szczepek from IMPB said, “The results of this work clearly show that the G protein and arrestin both contain a structurally very similar part with a homologous protein sequence, which binds and recognizes the receptor in a similar way”.
This paper has provided a new detailed insight into how our sense organs actually ‘sense’. Building on years of work, it furthers our understanding of the mechanisms by which GPCR receptor proteins interact with their partner proteins in the signal transduction chain. It’s well known that GPCRs play a vital role in physiological processes and in the development of diseases in the body. With at least one third of all currently available commercial medications directly acting on GPCRs, this work may have longer-term implications for the detailed understanding of protein receptors.
References
Crystal structure of a common GPCR-binding interface for G protein and arrestin, M. Szczepek et al., Nature Communications 5, September 2014.
Top image: The schema shows the rhodopsin signal transduction, from light absorption to switching off by binding arrestin.



