- Home
- News
- General News
- Fighting malaria...
Fighting malaria with X-rays
25-04-2018
Today, 25 April, is World Malaria Day. Considered as one of humanity’s oldest life-threatening diseases, nearly half the world population is at risk, with 216 million people affected in 91 countries worldwide in 2016. Malaria causes 445 000 deaths every year, mainly among children. The ESRF has been involved in research into Malaria since 2005, with different techniques being used in the quest to find ways to prevent or cure the disease.
Malaria in humans is caused by Plasmodium parasites, the greatest threat coming from two species: P. falciparum and P. vivax. The parasites are introduced through the bites of infected female Anopheles mosquitoes. They travel to the liver where they multiply, producing thousands of new parasites. These enter the blood stream and invade red blood cells, where they feed on hemoglobin (Hgb) in order to grow and multiply. After creating up to 20 new parasites, the red blood cells burst, releasing daughter parasites ready for new invasions. This life cycle leads to an exponential growth of infected red blood cells that may cause the death of the human host.
The research carried out over the years at the ESRF has aimed to identify mechanisms critical for the parasite’s survival in the hope of providing an intelligent basis for the development of drugs to stop the parasite’s multiplication and spread.
Understanding parasite activity
One such mechanism is the detoxification of heme, an iron-containing molecule that is released into the parasite upon hemoglobin degradation. The parasite neutralises heme by turning it into harmless hemozoin crystals, known as malaria pigment and built from heme dimers. Therefore, heme dimerisation and the subsequent crystallisation of the heme dimers are obvious goals for detailed experimental investigation.
In 2017, a research team from the Niels Bohr Institute, University of Copenhagen, the Weizmann Institute of Science in Israel and the Institute of Biology of the Humbolt University in Berlin, Germany, used the ESRF's ID16A beamline and the ALBA synchrotron in Barcelona, Spain, to investigate parasite-infected red blood cells. They used a combination of hard X-ray cryo-fluorescence microscopy and soft X-ray cryo-tomography. The sample - a single red blood cell - was immobilised by rapid cooling and then scanned with absorption tomography, allowing the scientists to map the cell in three dimensions. X-ray fluorescence mapping showed the chemical elements in the sample particularly the iron atoms which bind to heme and are abundant in hemozoin crystals. By overlaying the 3D structure of the cell and the X-ray fluorescence maps obtained, the distribution and concentration of chemical elements could be measured within various compartments of the parasite.
Tackling the resistance of antimalarial drugs
An essential goal of antimalarial drug discovery is how to overcome the problem of drug resistance. Using synchrotron nanochemical imaging on the ESRF’s former ID22 beamline (now ID16A/B), a team of researchers from the Université Lille 1, INSERM and the ESRF showed, for the first time, how antimalarial drugs are taken up and act in malaria-infected red blood cells.
The team was able to measure quantitatively the drug distribution inside the parasite, finding that it appears to selectively target the food vacuole where the parasite digests haemoglobin and where hemozoin is crystallised. The drug is found to cap the hemozoin crystals and in this way prevent the parasite to get rid of the toxic heme.
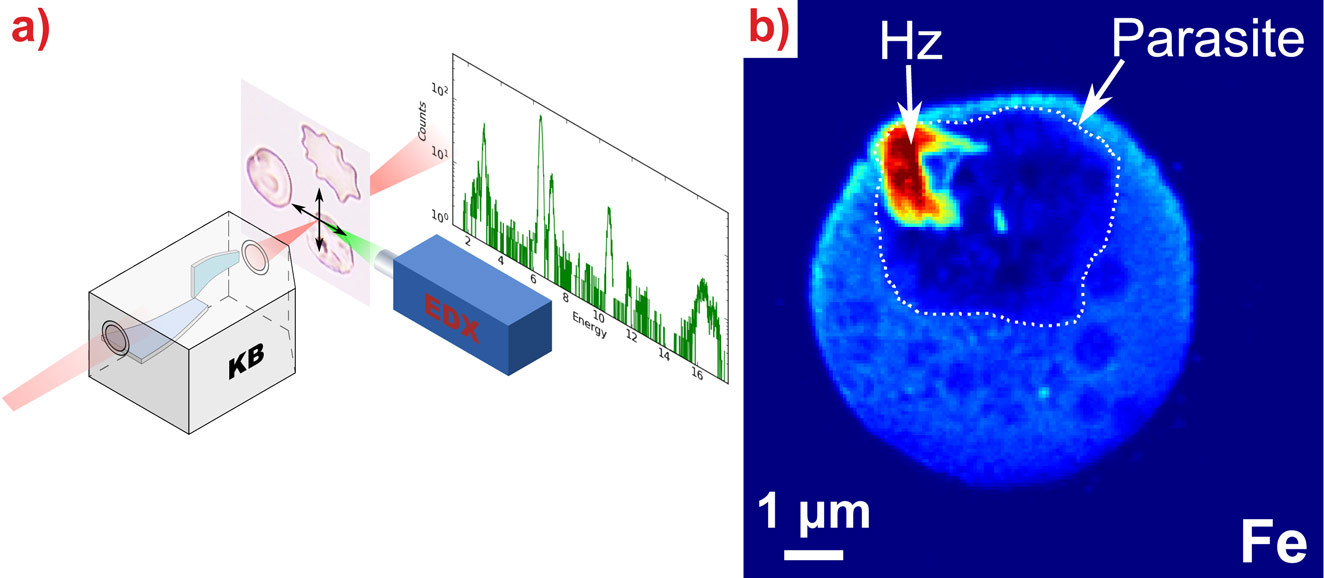 |
|
X-ray fluorescence microscopy of individual red blood cells. a) The sample is scanned through the nanofocus beam produced by the KB optics of the ID22NI (now ID16A/B) station while the X-ray fluorescence signal is being collected. b) Iron fluorescence signal of an infected red blood cell revealing sub-cellular features. The boundary of the parasite is delineated. The malaria pigment (hemozoin, Hz) has an increased iron content. |
 |
|
Inside the experimental hutch of the ESRF's ID16A nano-analysis beamline. ©Pierre Jayet |
Towards efficient vaccines
Ten years ago, the ESRF played a significant role in a major breakthrough in malaria research with a study published in Nature. Using X-ray crystallography, scientists studied the proteins on the surface of the Plasmodium parasite that enable it to interact and bind with proteins on the surface of red blood cells. For the first time, scientists were able to obtain a close-up view of where exactly on the surface of the cells the proteins interacted. This information was crucial to understanding how the parasite enters the cell. By examining the features of the binding process in atomic detail, it is possible to find ways of interfering with that process and further the development of drugs or vaccines to fight the illness.
For two decades, the ESRF has helped further malaria research thanks to the extremely bright X-rays and the development of new techniques to observe matter down to the nanoscale. The ESRF continues to develop its range of instrumentation and expertise for biochemical research. In November 2017, the ESRF inaugurated a Titan Krios cryo-electron microscope. This new equipment will complement the X-ray crystallography facilities onsite and offer the international structural biology community access to cutting-edge methods and instrumentation to address new biology and health challenges, including the quest to make malaria a disease of the past.
References
Biochemistry of malaria parasite infected red blood cells by X-ray microscopy, S. Kapishnikov et al. Sci Rep. 7, 802 (2017); doi: 10.1038/s41598-017-00921-2.
In situ nanochemical imaging of label-free drugs: a case study of antimalarials in Plasmodium falciparum-infected erythrocytes, F. Dubar et al Chem. Commun. 48, 910-912 (2012); doi:10.1039/C1CC16211J.
Structural basis for Duffy recognition by malaria parasite Duffy-binding-like domain. S.K. Singh et al, Nature 439, 741-744 (2006); doi:10.1038/nature04443.
Related articles
Toxoplasma's immunogenic balancing act explained
Exposing the ‘Achilles heel’ of dengue virus serotypes
Text by Kirstin Colvin
Top image: Anopheles albimanus mosquito engorged with blood while feeding on a human host. © James Gathany Content Providers: CDC [Public domain], via Wikimedia Commons



