- Home
- News
- Spotlight on Science
- Operando wide-angle...
Operando wide-angle X-ray scattering: a powerful tool for probing failures in CO electrolysis
23-11-2023
CO electrolysis is a key technology for achieving a carbon-neutral future; however, its large-scale application in membrane-electrode-assembly devices (MEA) faces durability challenges. Operando wide-angle X-ray scattering was used to uncover MEA degradation mechanisms, including gas diffusion electrode flooding and electrode contaminants during high-rate CO electrolysis.
CO electrolysis, the electrochemical conversion of carbon monoxide to produce valuable fuels and chemical feedstocks, presents a promising avenue for achieving carbon recycling in the transition towards a sustainable, carbon-neutral future [1]. However, a significant challenge for the widespread adoption of high-rate CO electrolysis is the limited durability of membrane-electrode-assembly (MEA)-based devices, stemming from issues that have remained unclear until now. This work combined operando wide-angle X-ray scattering (WAXS) measurements with detailed electrochemical characterisation to explore the degradation mechanisms of the MEA during high-rate and long-term CO electrolysis.
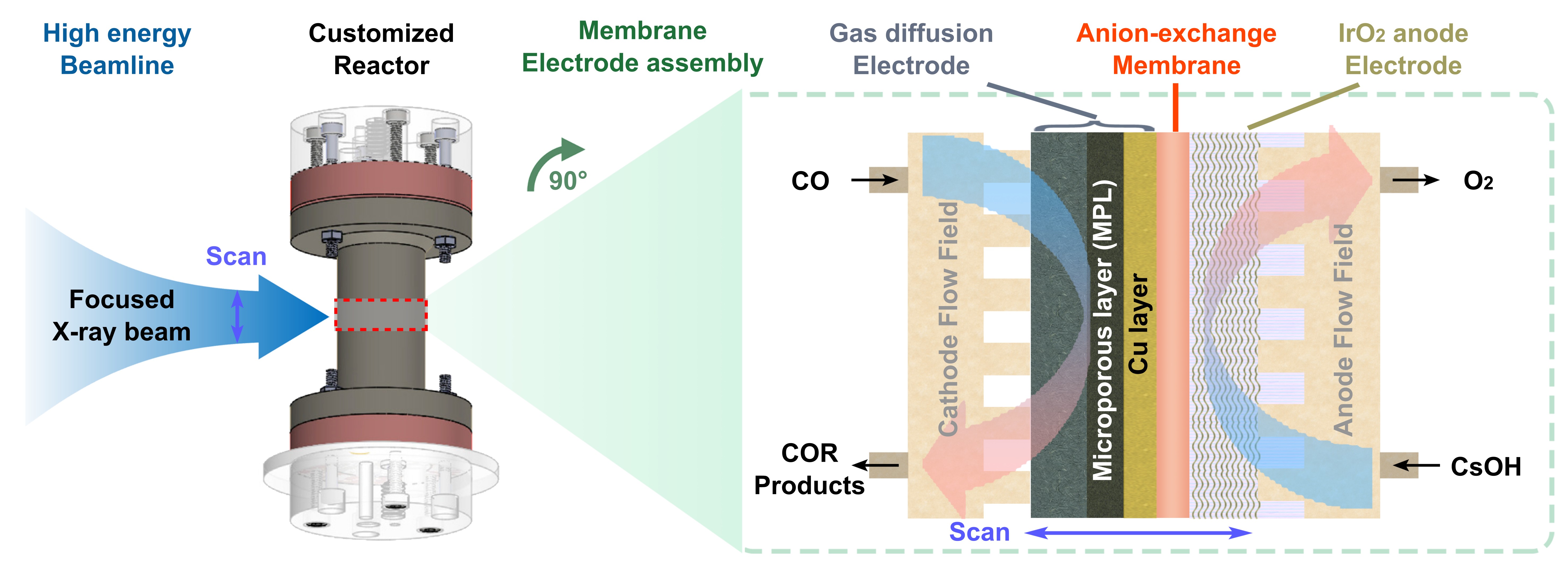
A customised synchrotron cell (Figure 1) was used to conduct CO electrolysis experiments at beamline ID31. During WAXS measurements, a high transmission and focused synchrotron X-ray beam (5 μm × 20 μm, vertical × horizontal) periodically scanned the MEA from the IrO2 anode through the membrane and all the way to the cathode flow field. Each scan takes 210 seconds to acquire 100 WAXS patterns across the MEA region. Therefore, each operando experiment produces substantial amounts of WAXS data in terms of both time and space, allowing for the tracking of real-time behaviour of the entire scanned MEA. From the intensity and background changes of characteristic signals in the WAXS mapping, the change in electrolyte content and the formation of new crystalline phases can be identified. Moreover, the gas products during CO electrolysis were measured by in-line gas chromatography.
Click image to enlarge
Fig. 1: Schematic diagram of the customised synchrotron cell and the MEA configuration for the operando CO electrolysis measurements by wide-angle X-ray scattering.
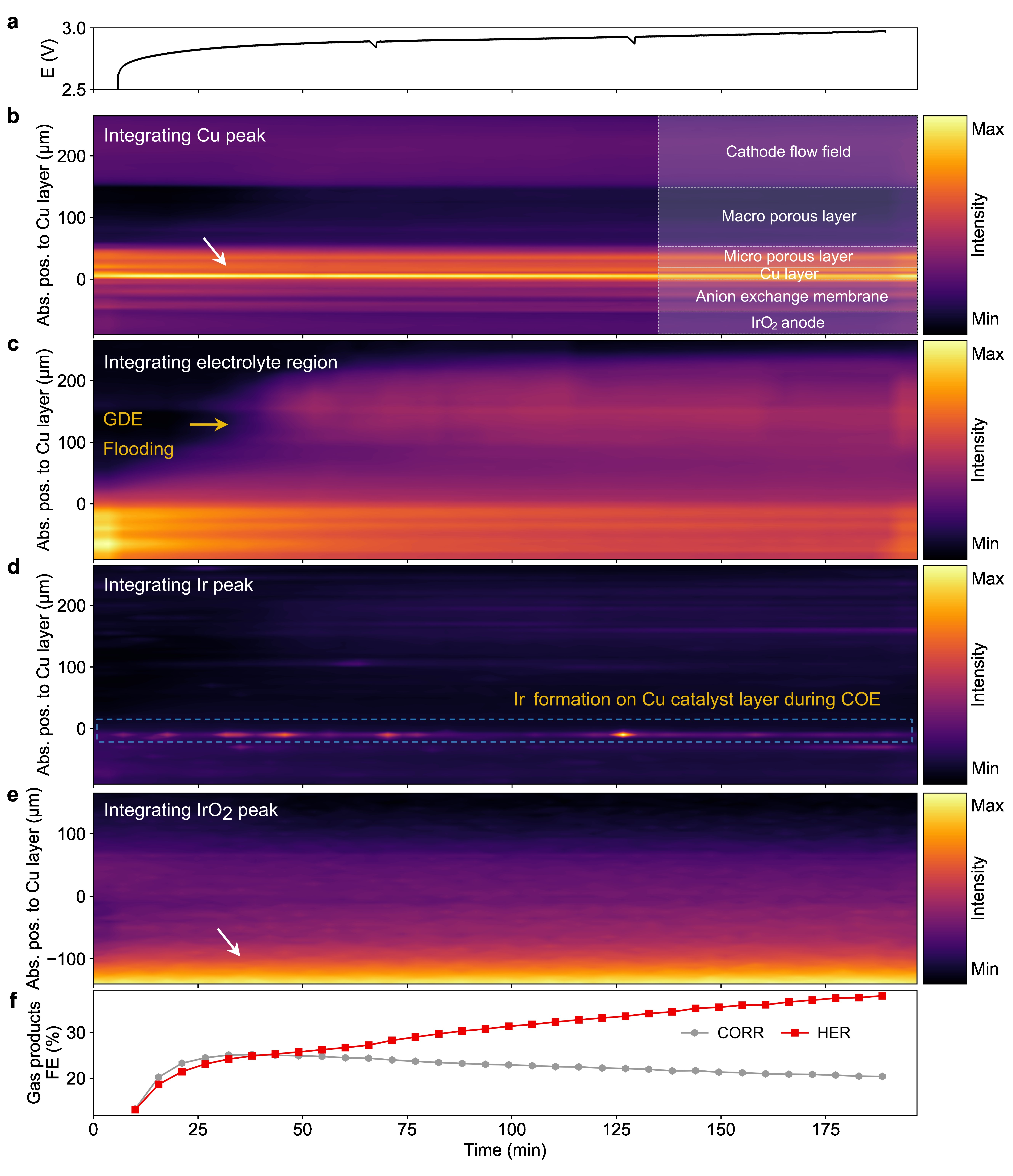
Figure 2 illustrates the typical MEA degradation behaviour during CO electrolysis. The cell voltage (Figure 2a) quickly increases in the first 30 minutes at a rate of ~ 5.5 mV min-1, and then increases much more slowly (~ 0.7 mV min-1) in the subsequent 150 minutes. This trend is in accordance with the increased water content in the gas diffusion electrode, where both macro- and micro-porous layers are flooded by water (Figure 2c). This gradual increase of water content in the gas diffusion electrode is hypothesised to be a reason for the Faradaic Efficiency shift towards the hydrogen evolution reaction (HER) (Figure 2f).
Click image to enlarge
Fig. 2: a) Cell voltage. b-e) The processed operando WAXS mappings by integrating corresponding fcc-structured Cu (111) peak (b), electrolyte (changes in background scattering (c), fcc-structured Ir (111) peak (d) and rutile-structured IrO2 (101) peak (e). f) Faradaic efficiencies (FE) of gas products for HER (i.e., H2) and CO reduction (including CH4 and C2H4) during CO electrolysis measurement (at 200 mA‧cm-2; anolyte of 0.1 M CsOH).
Furthermore, the WAXS mapping of the integrated Ir (111) and IrO2 (101) peaks, depicted in Figures 2d and e, reveals the presence of IrO2 exclusively on the anode side. In contrast, Ir appears to deposit on the Cu cathode during CO electrolysis. The Pourbaix diagram of the IrO2 indicates that IrO2 is unstable in alkaline conditions under oxidative potentials [2]. Thus, it is speculated that the Ir-based anode would slowly dissolve in anolyte with pH ~ 13 under oxidative potentials. Additionally, the underlying carbon on which IrO2 is supported may also be unstable under oxidative potentials, thus accelerating IrO2 dissolution. Once dissolved, it is then believed that the Ir-ions will cross over the anion exchange membrane and subsequently deposit on the cathode under a reductive potential. As a result, due to its high HER catalytic activity, the Ir contaminant induces a substantial increase in HER at the cathode.
To summarise, synchrotron WAXS results reveal that cathodic gas diffusion electrode flooding and Ir contaminants (crossover from anode) are two major issues causing excessive HER in a short-term testing period (< 3 hours). Based on this understanding, strategic measures were implemented to alleviate these issues. These include increasing the polytetrafluoroethylene content in the gas diffusion electrodes and using an alkaline stable Ni-based anode. Consequently, a 136-hour stability with a faradaic efficiency of CO reduction exceeding 70% relative to the hydrogen evolution reaction was achieved.
In conclusion, this work used operando WAXS to identify several degradation mechanisms of the MEA during high-rate and long-term CO electrolysis. The results provide guidelines to circumvent the durability challenges in the high-rate CO/CO2 electrolysis field, such as selecting the appropriate anode catalysts according to the reaction conditions and carefully monitoring the anodic oxidation reaction to avoid unexpected anode degradation.
Principal publication and authors
Identifying and alleviating the durability challenges in membrane-electrode-assembly devices for high-rate CO electrolysis, Q. Xu (a), S. Garg (a), A.B. Moss (a), M. Mirolo (b), I. Chorkendorff (a), J. Drnec (b), B. Seger (a), Nat. Catal. (2023); https://doi.org/10.1038/s41929-023-01034-y
(a) Surface Physics and Catalysis (Surf Cat) Section, Department of Physics, Technical University of Denmark, Kongens Lyngby (Denmark)
(b) ESRF
References
[1] M. Jouny et al., Nat. Catal. 2, 1062-1070 (2019).
[2] Z. Wang et al., Npj Comput. Mater. 6, 160 (2020).
| About the beamline: ID31 |
| Beamline ID31 is dedicated to the investigation of buried interfaces with high-energy X-rays using scattering techniques. With a focus on materials facilitating the energy transition, the beamline exploits the high flux and energy provided by ESRF-EBS to probe in operando reactions occurring in real devices (batteries, fuel cells, electrolysers, etc.), by combining wide- and small-angle X-ray scattering with other analytical tools such as impedance spectroscopy, gas analysis and chromatography. |