- Home
- News
- Spotlight on Science
- New anticancer compounds...
New anticancer compounds redistribute intracellular copper pools
24-02-2026
Copper-transporting molecules were synthesised to induce cancer cell death through alteration of intracellular copper distribution. X-ray fluorescence imaging of hepatic cancer cells at beamline ID21 revealed the intracellular distribution of copper, elucidating the mechanism of action of these compounds. This approach establishes a foundation for the development of novel therapeutic strategies targeting copper homeostasis in cancer.
Share
The challenge
Copper (Cu) is an essential trace metal for human health, yet it becomes toxic when present in its free form inside cells. To prevent this, cells tightly regulate intracellular copper levels through coordinated homeostatic mechanisms. Because copper also contributes to tumour development and progression, the disruption of copper homeostasis has emerged as a potential strategy for cancer therapy [1].
With the aim of developing a novel anticancer strategy, a family of compounds capable of binding Cu(I) ions – the major copper species present in cells – and transporting them across lipid membranes was synthesised [2]. Due to their ion-transporting properties, these compounds are referred to as ionophores. Although the total cellular copper content can be quantified by bulk analytical techniques, the visualisation of its intracellular distribution at the single-cell level remains challenging. X-ray fluorescence (XRF) imaging at beamline ID21 provides sub-micrometric spatial resolution and high elemental sensitivity, enabling the quantitative mapping of copper in frozen-hydrated cells.
The experiment
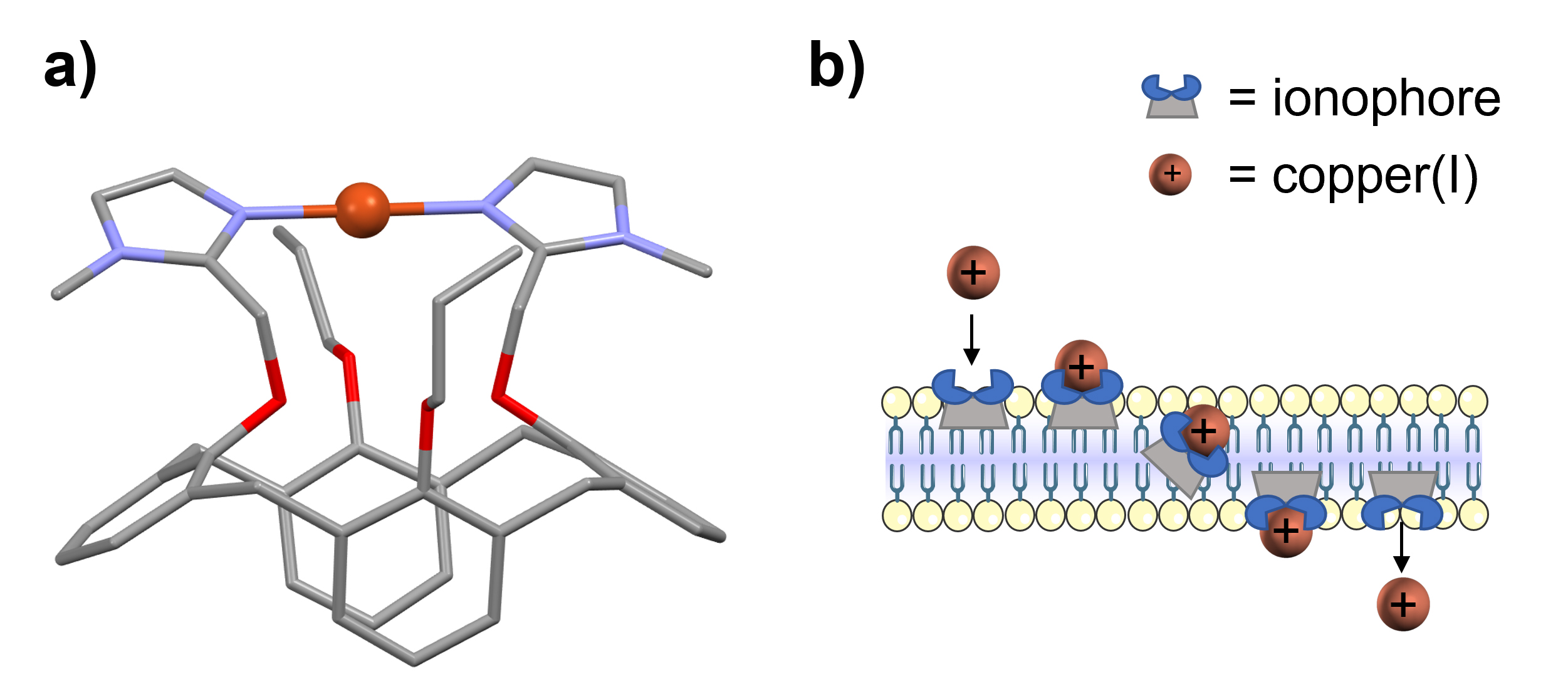
The anticancer activity of selected ionophores was tested on the HepG2/C3A hepatocarcinoma cell line, whose copper homeostasis has already been extensively studied. The mechanism of action of the most active ionophore, Cuphoralix (Figure 1a) [3], was investigated.
Click figure to enlarge
Fig. 1: Structure and mechanism of the Cu(I)-ionophore Cuphoralix.
a) X-ray crystal structure of the copper-bound synthetic copper ionophore Cuphoralix [3], consisting of two imidazole groups connected to a calix[4]arene platform. b) Schematic representation of its mechanism: the lipophilic ionophores selectively bind copper ions in the +1 oxidation state and transport them across membranes. This transport property induces copper poisoning and consequent cell death in cancer cells.
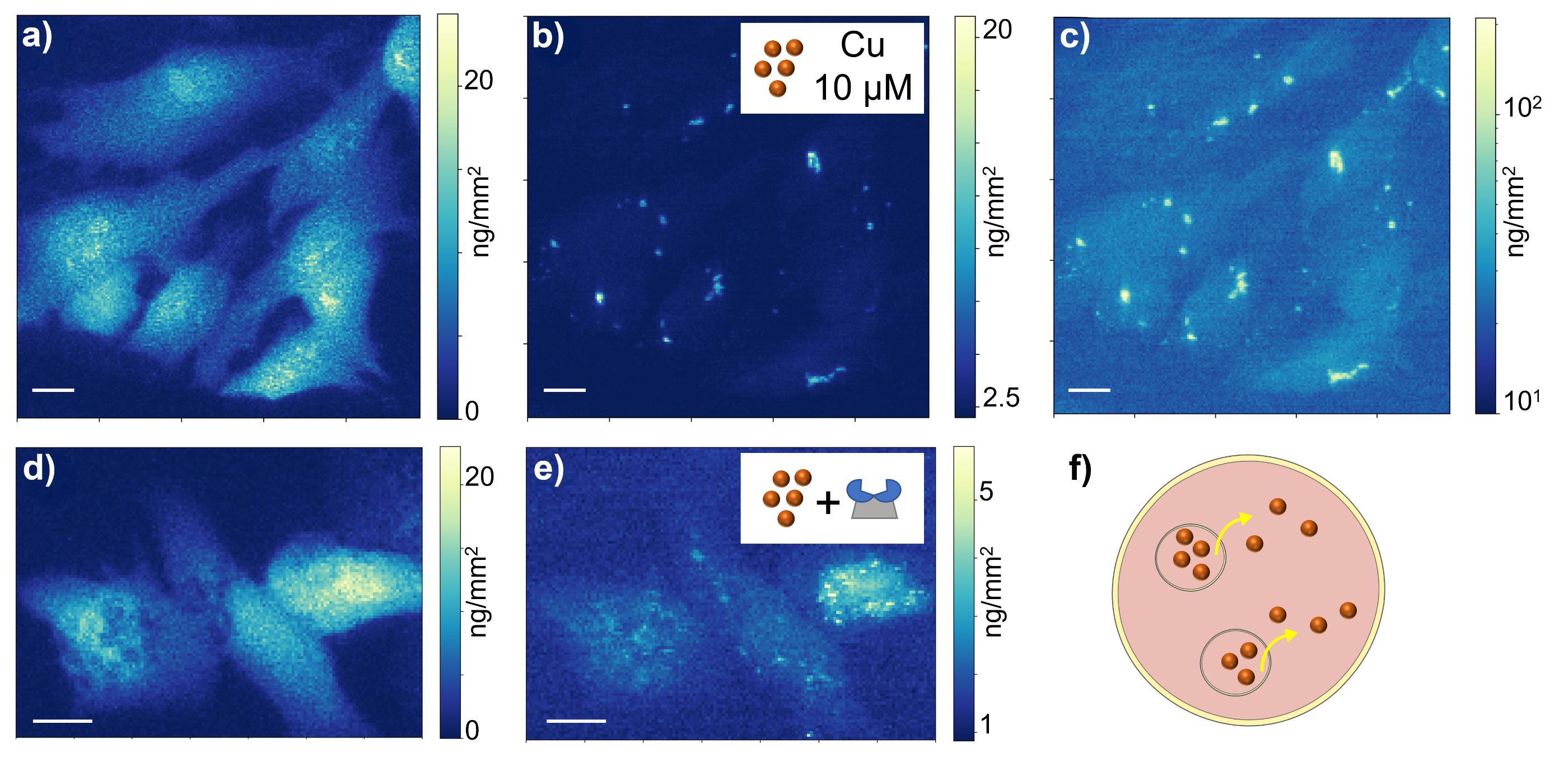
XRF hyperspectral images of HepG2/C3A cells exposed to a non-toxic concentration of Cu (10 µM) were acquired at ID21’s scanning X-ray microscope, allowing for the determination of the distribution of physiological elements such as potassium (Figure 2a), which delineates the cell area. Copper was observed as highly localised spots of elevated concentration (up to 20 ng/mm2, Figure 2b), while only a weak Cu signal corresponding to low concentration was detected outside these spots in the cellular cytosol (Figure 2c). This distribution is consistent with the formation of storage vesicles that confine excess copper to prevent toxic effects.
To assess the modifications of copper homeostasis induced by the ionophore, the latter was co-administered with Cu (10 µM). XRF images revealed a markedly different copper distribution (Figure 2e), in which the metal was no longer confined in vesicles but diffusely distributed throughout the cell.
Click figure to enlarge
Fig. 2: XRF imaging reveals copper redistribution in hepatic cancer cells.
False-colour XRF maps of potassium (a,d) and copper (b,c,e) in hepatic cancer cells exposed to 10 µM copper (a,b,c) or to 10 µM copper in conjunction with Cuphoralix (d,e). Under copper-only conditions (b,c), the metal accumulates in concentrated vesicles. Logarithmic scaling (c) reveals that vesicular copper concentration is approximately one order of magnitude higher than in the surrounding cytosol. In the presence of Cuphoralix (e), vesicular storage is hindered, inducing the release of copper from intracellular vesicles to the cell cytosol, as illustrated in schematic (f). Scale bars: 10 μm.
Inductively coupled plasma mass spectrometry measurements demonstrated that the average copper content per cell remained unchanged with or without the ionophore. The results indicate that the anticancer activity of Cuphoralix relies on the redistribution of intracellular copper rather than on increased cellular uptake.
The impact
These findings suggest a potential route toward new anticancer therapies based on the disruption of copper homeostasis. The results demonstrate that the redistribution of intracellular Cu(I), the predominant copper form in mammalian cells, leads to cancer cell death. Cu(I) trafficking pathways therefore represent promising therapeutic targets, supporting the development of strategies based on selective modulation of intracellular metal distribution.
|
Principal publication References |
|
About the beamline: ID21 Following a recent upgrade, ID21 now hosts two scanning X-ray microscopes. A newly installed nanoscope (nano-SXM) is optimised for high-resolution (down to 120 nm), high-speed nano-XRF mapping and nano-XAS, offering enhanced performance over the previous microscope. The original microscope has been retained and is now dedicated to larger samples, using micrometric and submillimetric beams. Both microscopes operate under vacuum and support room temperature and cryogenic measurements. |