- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2012
- X-ray imaging
- 3-D bone ultrastructure resolved by phase nanotomography
3-D bone ultrastructure resolved by phase nanotomography
Bone tissue was imaged at the sub-cellular level using synchrotron radiation phase nanotomography. Previously, images at the ultrastructural level have only been available with electron imaging techniques, such as scanning or transmission electron microscopy, thus in 2D. In this study, bone ultrastructure was imaged in 3-D with a 60 nm voxel size. Several ultrastructural properties could be visualised in 3-D for the first time: the collagen fibre orientation, the mineralisation on the nanoscale, and the imprint of the cellular network.
Bone is a hierarchically organised material with remarkable capabilities to maintain and repair itself. However, due to aging or disease, bone becomes fragile, leading to fracture. Understanding the mechanisms determining bone strength and fragility is the prerequisite for advances in bone tissue engineering and disease treatment. For example, the role of the osteocyte, a bone cell embedded in the bone matrix, in sensing damage and converting forces to biological signals has been highlighted. Despite recent advances, a clear picture of the 3-D nano-organisation of bone has not yet been established due to lack of suitable 3-D imaging techniques [1-3].
 |
|
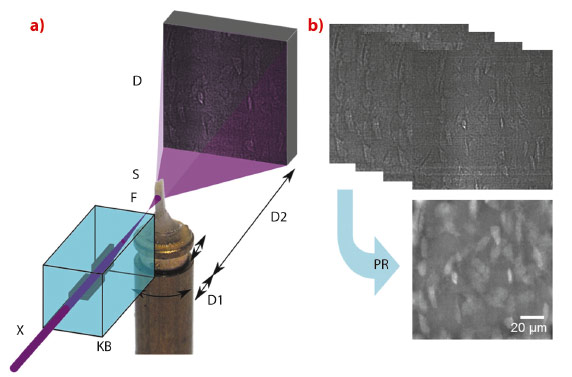
Fig. 71: a) Schematic of experimental setup. The X-ray beam (X) is monochromatised and focused into a focal spot (F) by multilayer coated KB optics. The sample (S) is positioned on a translation-rotation stage downstream of the focus and imaged onto a stationary detector. Due to the resulting divergent beam, different spot-sample distances (D1) and different free space propagation distances (D2) imply different magnification factors on the detector. b) Images were recorded at four focus-to-sample distances over a complete turn of the sample at 2999 projection angles. The images were used to reconstruct the phase shift at each angle (phase retrieval, PR), which was used as input to a tomographic reconstruction algorithm to reconstruct the 3-D local mass density. |
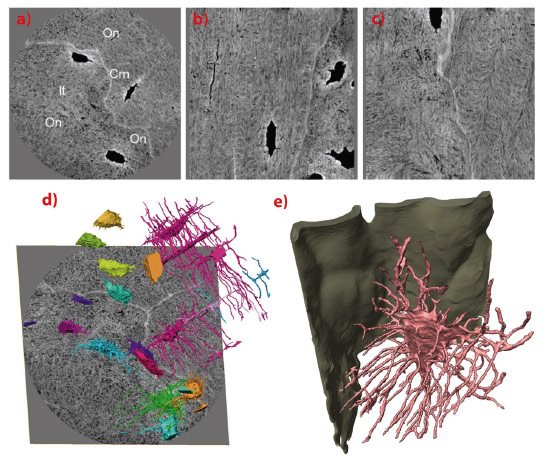
Magnified X-ray phase nanotomography was performed at the nano-imaging beamline end-station ID22NI (Figure 71). 3-D maps were obtained of the mass density within human cortical bone samples at 60 nm spatial resolution (Figure 72). The images reveal the organisation of the lacunae, which contain the osteocytes in living bone, and the canaliculi through which they connect in a large region. The cement line, separating bone tissue of different age, could also be studied in 3-D for the first time (Figure 72). In this study we were able to measure the relative electron density directly, and we found that the cement line is hypermineralised. For the first time, the collagen organisation in the compact bone matrix could be directly observed in 3-D.
 |
|
Fig. 72: a) Transverse, b) frontal and c) sagittal slices through the images reconstructed from phase data. Grayscale is proportional to local density. Osteocyte lacunae (Lc) and canaliculi (Ca) can clearly be seen. The heterogeneous organisation of the matrix by mineralised collagen fibres can also be distinguished. The cement line (Cm), separating osteonal (On) and interstitial (It) tissue, can clearly be distinguished as more mineralised than the surrounding matrix. Tissue close to osteocyte lacunae is also hypermineralised. d) Rendering of osteocyte lacunae and canaliculi in the whole imaged volume overlayed on the bottom slice shown in grayscale. Colours correspond to connected components and grayscale to mass density. e) Enlargement of a lacuna showing the interaction between the canaliculi (pink) and the cement line (green), and branching of the canaliculi. |
The information content in the 3-D images is exceptionally high due to the high spatial resolution and large field of view. Furthermore, the technique requires very little sample preparation, thus avoiding possible unintentional altering of structures. The sensitivity is exceptional and appears to outperform attenuation based full-field X-ray microscopy and ptychography [2,3]. In contrast to scanning techniques, it is possible to keep acquisition times short (less than two hours per sample) thus facilitating the imaging of series of samples. We believe this method will have a significant impact in osteology since it gives access to information unavailable with other techniques, while being fast and reproducible. Therefore, this work paves the way for new studies to gain a better understanding of bone function, remodelling, and ultimately to new strategies in bone repair and the management of bone related diseases.
Principal publication and authors
M. Langer (a,b), A. Pacureanu (a,b,c), H. Suhonen (b), Q. Grimal (d), P. Cloetens (b) and F. Peyrin (a,b,e), PLoS ONE 7, e35691 (2012).
(a) CREATIS, CNRS 5220, INSERM U1044, INSA Lyon, Université de Lyon (France)
(b) ESRF
(c) Current address: Centre for Image Analysis & SciLifeLab, Uppsala University (Sweden)
(d) UPMC Univ Paris 06, CNRS UMR 7623, LIP, Paris (France)
(e) Labex PRIMES, Université de Lyon (France)
References
[1] L.F. Bonewald, J. Bone Miner. Res. 26, 229-238 (2011).
[2] M. Dierolf et al., Nature 467, 436-439 (2010).
[3] A. Pacureanu, M. Langer, E. Boller., P. Tafforeau and F. Peyrin, Med. Phys. 39, 2229-2238 (2012).



