- Home
- Users & Science
- Find a beamline
- Structural biology
- UPBL10 and MASSIF
UPBL10 and MASSIF
A description of the evolution of the Structural Biology beamlines in the context of the ESRF upgrade
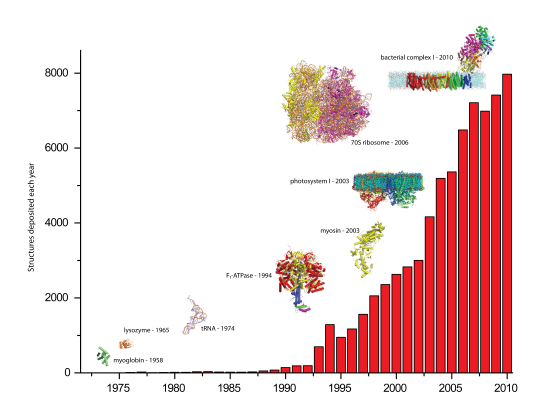
Structural Biologists are tackling ever more ambitious projects, for example more complex membrane proteins and larger macromolecular assemblies (Figure 1). Such systems often show considerable inter and intra crystal variation in diffraction quality. Sample evaluation prior to data collection, already widespread in Macromolecular Crystallography (Figure 2), will thus become more crucial as will data collection facilities optimised for the collection of diffraction data from crystals that are very small and/or diffract to low resolution (i.e. dmin 4 – 5 Å).
Figure 1. An illustration of the increase in numbers of deposited macromolecular structures and the size of the complexes studied in MX since the solution of the first enzyme structure in 1965.
UPBL10 (MASSIF - Massively Automated Sample Selection Integrated Facility) will be a unique resource, based on 2nd generation automation for MX experiments, designed to maximise the chances of a successful conclusion to these ambitious projects. The hub of the facility is a sample evaluation and sorting facility. The most suitable crystals from which to collect data will be distributed from this hub to the data collection facilities that form an integral part of this proposal as well as to existing facilities on ID29 and ID23.
Data collection trends at the ESRF MX beamlines show clearly that automation has led to a fundamental change in the way that MX data are collected. With the introduction of the automatic sample changer (SC3) at the ESRF MX beamlines in 2005, a dramatic increase in the number of samples that were tested for diffraction quality before any full data collections were carried out was observed. Automation has had a spectacular effect on the scientific output of the ESRF MX beamlines. Over the past seven years there has been a more than three-fold increase in the number of structures elucidated using data collected at the ESRF. Although the output has increased - MX beamlines now produce over a quarter of the publications from the ESRF - this has not been at the cost of quality. The scientific impact of the experiments remains extraordinarily high, with around 20% of publications appearing in journals with an Impact Factor greater than 10. Automation of synchrotron beamlines thus not only increases scientific output but it also maintains the high-level impact of the science performed.
Figure 2. The MASSIF beamlines - three highly intense (>1012 ph/s) beams (two 100 μm (blue) and one 10 μm) are provided for 2 experimental hutches. The beamlines will be optimised for the automated high throughput screening of large numbers of samples.
In essence, UPBL10 will provide an upgraded set of beamlines regenerating ID14 A&B on ID30. The project consists of a suite of beamlines. Three end-stations will provide facilities for different types of sample evaluation: testing the diffraction resolution limit, locating the best part of a crystal on which to perform data collection, detecting the presence of anomalous scatterers, screening for bound inhibitors and ligands, screening in crystallisation plates - integrating and building on the developments made with DNA and EDNA. The screening stations directly address a fundamental problem in modern Structural Biology – that of inter and intra sample variability and will have different beam sizes (two stations with 80 μm beam diameter and one microbeam station with a beam-size of 10-20 μm in diameter).
The development of the automation has begun on ID14-2 with the installation of a GRob robot to pilot the sample changing robotics for MASSIF (Figure 3). Other developments are being carried out on the other Structural Biology beamlines such as full diffraction mapping of samples on ID23-2 (Figure 4).
Figure 3. The GRob mounted on ID14-2. ID14-2 is piloting the MASSIF beamlines - a new diamond monochromator (Laue 110) has been installed and the robotics are being tested for installation on MASSIF.
Identifying the “best” crystal is only the beginning of a successful MX experiment. UPBL10 will also provide a tuneable end-station, from an independent canted source, to provide a facilitiy optimised for the data collection requirements of the crystals identified as being suitable. Exploiting the potential of a high β-source, the end-station will have a relatively large beam (~ 200 μm) with an X-ray collimation system able to tailor the size and shape of the X-ray beam to that of the sample. This will increase signal-to-noise ratios, thus maximising the number of reflections observed and reducing the exposure times required for the collection of high quality data. The low divergence of the beam will also be extremely useful in data collection from crystals with large unit cells (>1000 Å). The end-station could also be specialised in low resolution studies (dmin to ~1000 Å) and be optimised for the measurement of very weak anomalous scattering signals. Additionally, there will be a permanently mounted on-line micro-spectrophotometer and humidity control device.
UPBL10 is to be placed on ID30 integrating the facility with the current SB beamline ID29 and increasing communication with the French Beamline for the Investigation of Proteins (FIP) at BM30. Additionally BM29, located between the two beamlines, is to be converted to a solution scattering beamline dedicated to biological samples (Figure 3) replacing ID14-3. The integrated resources centered around ID29 and ID30, together with the state of the art beamlines ID23-1 and ID23-2, will provide a set of beamlines for the collection of the highest quality diffraction data from the wide variety of samples that will arise from Structural Biology studies of challenging biological problems. It will also be able to cope with the projected three to five fold increase in sample numbers arising from the search for appropriate crystals from which to collect data.
Construction of the optics and experimental hutches for MASSIF (ID30A) has been finished and commissioning of equipment is currently under way.

Figure 4. Hutch construction finished at ID30.