- Home
- News
- Spotlight on Science
- Scintillating nanoparticles...
Scintillating nanoparticles enhance radiotherapy efficacy in preclinical models of glioblastoma
12-03-2021
A study performed at the biomedical beamline ID17 demonstrates that nanoscintillators enhance the efficacy of synchrotron radiotherapy for glioblastoma microtumours and prolong survival in a preclinical model of aggressive glioblastoma.
Share
Radiation therapy remains a major part of the anti-cancer arsenal, with more than 50% of cancer patients undergoing radiotherapy, however, its efficacy still needs to be improved. Indeed, due to radiation toxicity and a lack of specificity for tumour tissues, delivering a high dose of X-rays with tolerable toxicity to the surrounding tissues remains a clinical challenge that limits the treatment of cancer. This is exemplified in multiform glioblastoma, the most common type of primary brain cancer in adults.
Glioblastoma has a dismal prognosis: the median survival does not exceed 15 months despite the aggressive standard of care, which involves a maximal safe surgical resection followed by radiotherapy and chemotherapy. For glioblastoma, the radiation dose that would be necessary to sterilise the tumour highly exceeds the dose that can be tolerated by healthy surrounding tissues, therefore explaining the poor outcome of radiotherapy. Radiation dose-enhancement is therefore one of the strategies to improve the effects of radiotherapy by increasing the contrast between the dose delivered to the tumour and the dose received by healthy tissues – crucial to improve glioblastoma prognosis.
Recent studies have demonstrated the ability of scintillating nanoparticles, a specific type of high-Z element nanoparticles, to potentiate radiotherapy [1,2]. Nanoscintillators are able to down-convert ionising radiation, such as X-rays, into visible light. Whereas scintillating materials are usually used for detection purposes, nanoscintillators have gained interest for their potential as radiotherapeutics due to various possible contributions that can be activated upon radiotherapy. The success of radiation dose-enhancement relies on a higher absorption cross-section for high-Z elements than tissues, which induces a local overproduction of photoelectrons and Auger electrons. Assuming that the high-Z elements are accumulated in the tumour prior to radiotherapy, they will create a dose-enhancement effect in the tumour. Therefore, this modality is particularly promising when using X-rays with energies below 100 keV as the absorption cross-section difference between nanoparticles and tissues will be maximised and the photoelectric effect will dominate the interactions. Synchrotron radiation is a unique tool to investigate the physical processes accountable for the therapeutic improvement, as it allows the delivery of monochromatic radiation with tuneable energy in the optimal domain. The biomedical beamline ID17 is a valuable tool for this purpose, and is currently unique in the world.
Researchers using monochromatic synchrotron radiation therapy and computed tomography at beamline ID17 investigated the particular radiation dose-enhancement effect induced by nanoscintillators and, more particularly, by LaF3:Ce nanoparticles. A first study was performed in vitro on 3D models of glioblastoma tumours. After showing that LaF3:Ce nanoscintillators were well-accumulated by microtumours and presented a low toxicity profile in vitro, the efficacy of radiotherapy in the presence of nanoscintillators was investigated. In order to unravel the physical mechanism accountable for this improvement, the necrosis obtained after radiation therapy delivered at various X-ray energies was measured. By revealing a correlation between the extent of necrosis and the absorption cross-section of the LaF3:Ce nanoparticles, the radiation dose-enhancement effect was unequivocally identified as accountable for the improved radiotherapy efficacy.
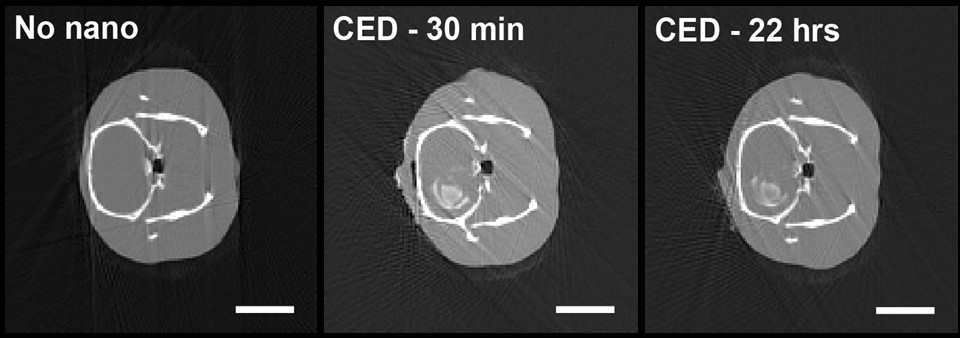
Fig. 1: Representative computed tomography images of a control model (no nanoparticles), as well as two models that received an intratumoural injection of LaF3:Ce nanoscintillators 30 minutes or 22 hours before imaging. Scale bar = 10 mm.
A preclinical study was then performed in a highly aggressive model of syngeneic glioblastoma (F98 cells). This model was chosen as it recapitulates the aggressivity, invasiveness and treatment resistance observed in the human glioblastoma. After demonstrating that LaF3:Ce nanoscintillators presented no limiting toxicity when injected intravenously or intratumourally and were well-eliminated, their ability to improve survival was studied. Interestingly, it was demonstrated that a combination of LaF3:Ce nanoscintillators (Figure 1) and 15 Gy of 50 keV radiotherapy significantly improved survival. Further investigation revealed that the results would be further improved by perfecting the specific accumulation of the nanoscintillators in tumour cells, emphasising the potential of nanoscintillator-induced radiation dose-enhancement effects to improve the clinical effects of radiotherapy.
Principal publication and authors
Radiation Dose-Enhancement Is a Potent Radiotherapeutic Effect of Rare-Earth Composite Nanoscintillators in Preclinical Models of Glioblastoma, A.-L. Bulin (a), M. Broekgaarden (a), F. Chaput (b), V. Baisamy (a), J. Garrevoet (c), B. Busser (d), D. Brueckner (c), A. Youssef (a,e), J.-L. Ravanat (e), C. Dujardin (f), V. Motto-Ros (f), F. Lerouge (b), S. Bohic (a), L. Sancey (d), H. Elleaume (a), Adv. Sci. 7, 2001675 (2020); https://doi.org/10.1002/advs.202001675.
(a) Synchrotron Radiation for Biomedical Research (STROBE), INSERM-University Grenoble Alpes, Grenoble (France)
(b) École Normale Supérieure de Lyon, Lyon (France)
(c) Deutsches Elektronen-Synchrotron DESY, Hamburg (Germany)
(d) Institute for Advanced Biosciences, INSERM-CNRS-University Grenoble Alpes, Grenoble (France)
(e) CEA Grenoble, Grenoble (France)
(f) Institut Lumière Matière, Lyon (France)
References
[1] A.-L. Bulin et al., J. Phys. Chem. C 117, 21583-21589 (2013).
[2] A.-L. Bulin et al., Nanoscale 7, 5744-5751 (2015).



