- Home
- News
- Spotlight on Science
- Nanoscale X-ray...
Nanoscale X-ray imaging shows photodynamic therapy boosts chemotherapy in pancreatic cancer
10-11-2025
X-ray fluorescence-based imaging at beamline ID16A was used to map how chemotherapy drugs distribute inside pancreatic cancer cells. Photodynamic therapy increased the intracellular availability of the drug. The results support PDT as an adjuvant strategy and provide a rationale for future clinical trials.
Share
The challenge
Pancreatic cancer is among the deadliest malignancies, with 1-year survival below 20%. Because diagnosis is often late, many patients cannot undergo surgery and rely on palliative chemotherapy. Regimens such as FOLFIRINOX can extend survival to around 11 months, but toxicity limits their use, creating a need for combinations that are more effective and better tolerated.
Photodynamic therapy (PDT) uses photosensitisers that accumulate in tumours and, upon local light activation, generate reactive oxygen species (ROS) that kill tumour cells while sparing surrounding tissues. Fibres for light delivery can be placed endoscopically, making PDT clinically feasible for pancreatic lesions.
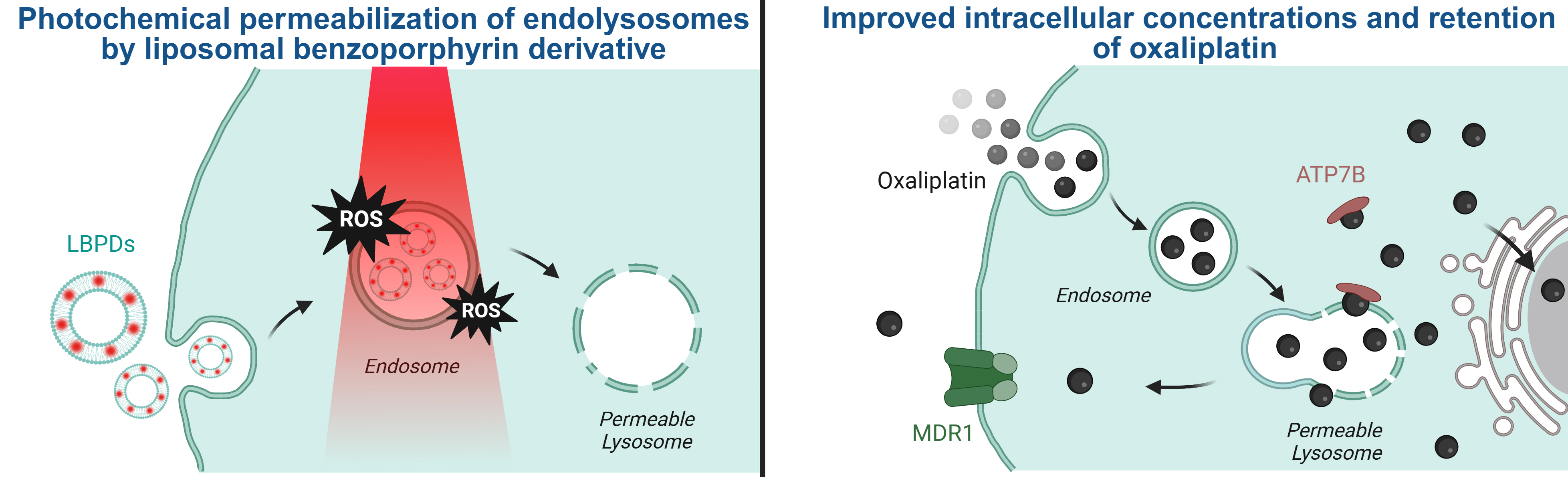
Preclinical investigations demonstrated that combining PDT with oxaliplatin was beneficial, but the underlying mechanisms were unclear. Owing to its hydrophilic nature, oxaliplatin is likely internalised via endocytosis and sequestered within lysosomes – the cell’s garbage disposal system – restricting its bioavailability. It was therefore hypothesised that PDT could permeabilise lysosomal membranes and release endocytosed oxaliplatin into the cytosol (the aqueous component of the cytoplasm), increasing its effectiveness (Figure 1).
Click figure to enlarge
Fig. 1: Schematic representation of the increased intracellular accumulation of oxaliplatin. Left: The photosensitisers in their lipid nanocarriers (liposomal benzoporphyrin derivative, or LBPDs) are taken up by endocytosis into cancer cells. Irradiation of the cancer cells generates reactive oxygen species (ROS), permeabilising the lysosomes. Right: Subsequent administration of oxaliplatin leads to similar endosomal uptake, but the endosomes then fuse with the permeable lysosomes. This results in increased intracellular platinum levels (although sequestration by ATP7B copper chelating proteins or efflux via multidrug resistance protein 1 (MDR1) may occur).
The experiment
Using the clinically applied benzoporphyrin derivative as a carrier, synergy between PDT and oxaliplatin was confirmed in both conventional and 3D cell culture models, as well as in mouse models of pancreatic cancer. When endocytosis was inhibited, oxaliplatin became less effective, showing that endocytosis is required for drug uptake and that part of the drug becomes trapped in lysosomes.
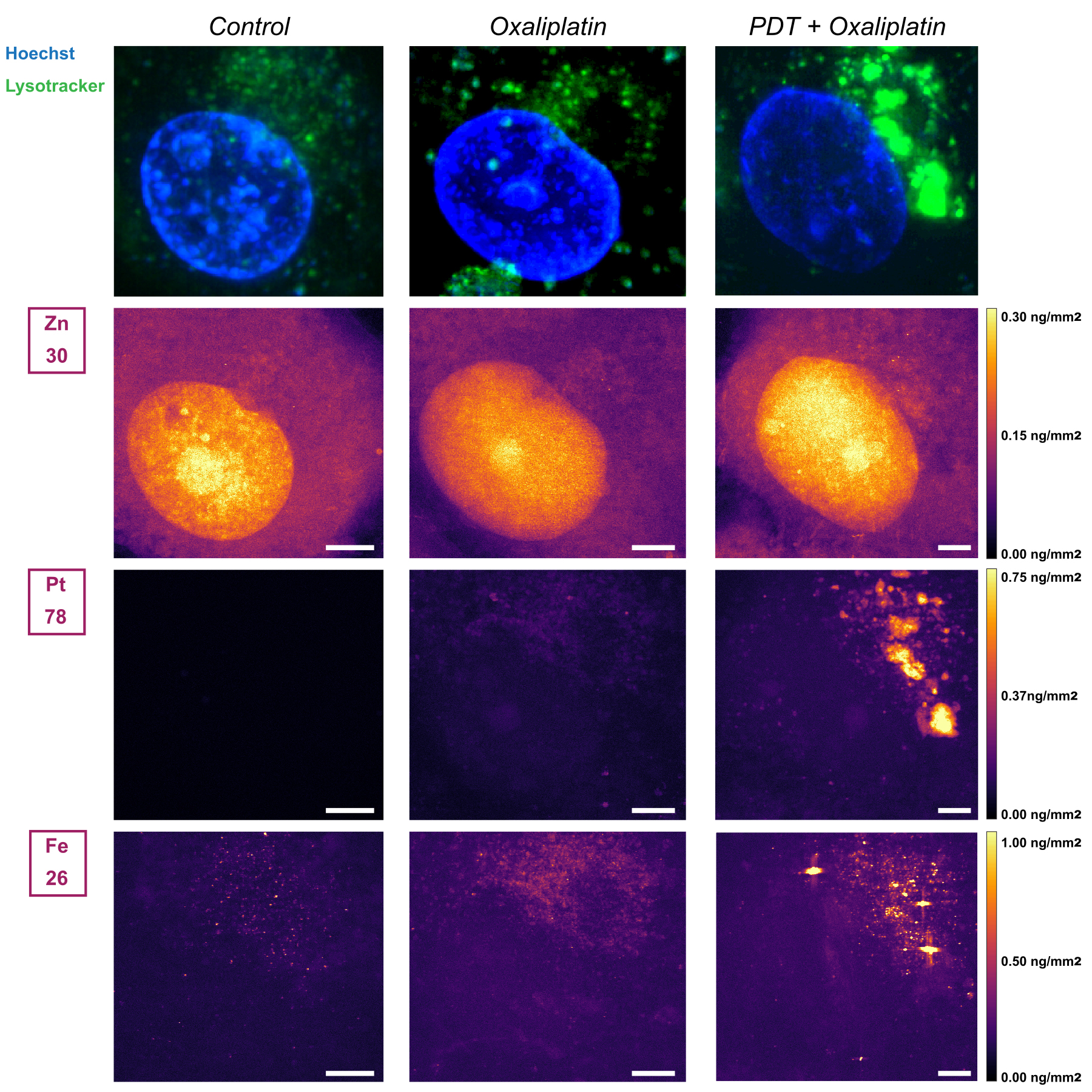
To visualise this process directly, X-ray fluorescence imaging of cryo‑preserved cells was performed at the cryo-nanoprobe beamline ID16A (Figure 2). After oxaliplatin alone, platinum was detected in discrete foci that overlapped with lysosomal staining, confirming lysosomal sequestration. After PDT, intracellular platinum increased markedly; most of it still colocalised with lysosomes, but additional signals suggested binding to copper‑handling proteins. Platinum in the nucleus remained very low, indicating that the classical DNA‑damage mechanism may not be the only relevant pathway.
Click figure to enlarge
Fig. 2: Alterations in intracellular oxaliplatin accumulation by PDT as determined by X-ray fluorescence-based elemental imaging at beamline ID16A. Cells were either left untreated, treated with 10 µM oxaliplatin for 24h, or treated with PDT (0.5 J/cm2) followed immediately by oxaliplatin exposure at 10 µM. X-ray fluorescence imaging was then performed, from which zinc (Zn, nucleus), platinum (Pt), and iron (Fe) concentrations (as a possible indication of ferroptosis) were extracted.
Overall, this study shows that PDT reshapes the intracellular localisation and effective concentration of oxaliplatin and highlights endocytosis as a key step for highly hydrophilic chemotherapeutics. These findings justify clinical studies testing PDT as an add‑on to FOLFIRINOX in pancreatic cancer.
Principal publication
Photochemical internalization with verteporfin-liposomes enhances oxaliplatin retention and efficacy in models of pancreatic cancer, N.M. Carigga Gutierrez et al., J. Controlled Release 387, 114201 (2025); https://doi.org/10.1016/j.jconrel.2025.114201
| About the beamline: ID16A |
| The ID16A cryo-nanoprobe beamline is designed for quantitative 3D characterisation at the nanoscale of the morphology and the elemental composition of specimens in their native state. It addresses research questions in metallo-biology, neurosciences, biomineralisation, and advanced materials characterisation. The beamline provides a high-brilliance beam focused down to a few tens of nanometres to perform coherent imaging techniques, X-ray fluorescence microscopy, and nanotomography. All measurements can be performed under cryogenic conditions to preserve the biological samples close to their native hydrated state and reduce radiation damage. |