- Home
- News
- Spotlight on Science
- Nanoholotomography...
Nanoholotomography tracks fertiliser nanoparticles in living plants
17-12-2025
Overuse of agricultural fertilisers is driving eutrophication and long-term ecosystem degradation. X-ray nanoholotomography at beamline ID16B was used to image fertiliser nanoparticles in living plant tissues at sub-200 nm resolution, providing insight into foliar nutrient uptake and transport to guide the development of more sustainable fertiliser strategies.
Share
The challenge
The global use of mineral fertilisers in agriculture has increased dramatically over recent decades, enabling major gains in agricultural productivity but at considerable environmental cost. Current fertiliser application rates exceed the Earth system’s safe operating limits for both nitrogen and phosphorus cycling, contributing to soil degradation, freshwater eutrophication, and downstream ecological damage.
For example, the annual flow of phosphorus from agricultural soils into freshwater systems is currently estimated at approximately 22 Tg, around twice the proposed safe limit [1]. These losses reduce nutrient-use efficiency and pose long-term risks to aquatic and marine ecosystems.
Despite the scale of these impacts, the physical pathways by which nutrients enter plants and are transported within living tissues remain incompletely understood. Conventional fertilisation relies predominantly on uptake through the root–soil interface, where nutrients are subject to immobilisation, leaching, and microbial transformation.
Foliar fertilisation has emerged as a promising alternative approach, offering the potential to bypass inefficient soil pathways and deliver nutrients directly through leaves. In particular, the use of engineered nanoparticles has been proposed as a means of enhancing nutrient delivery and retention [2]. However, the translocation mechanisms of such particles within living plant tissue remain poorly characterised, largely due to the lack of suitable non-destructive analytical tools.
Advanced three-dimensional imaging methods are required to visualise nanoparticle transport in vivo while preserving tissue integrity. Classical nano-resolution X-ray imaging techniques typically impart radiation doses that are incompatible with living organisms, necessitating chemical fixation or dehydration. By contrast, nanoholotomography at high X-ray energies offers substantially improved dose efficiency.
At the ESRF’s ID16B beamline, a lensless imaging geometry combined with the high coherence of the Extremely Brilliant Source enables three-dimensional phase-contrast imaging of living plant tissue at sub-200 nm resolution without inducing immediate structural damage.
The experiment
The uptake and localisation of fertiliser nanoparticles in living plant leaves were investigated using X-ray nanoholotomography at ID16B. Three-dimensional imaging was performed on intact leaves maintained in a hydrated, living state, allowing nanoparticle behaviour to be examined under physiologically relevant conditions. Sub-100 nm poly(acrylic acid)-based manganese dioxide nanoparticles (nHAA–MnO2) were applied to barley leaves and imaged at successive time points (Figure 1).
Click image to enlarge
Fig. 1: Nanoholotomography images of MnO2 fertiliser nanoparticles in a living barley leaf after induced aggregation. a) Virtual 2D slice of the data showing mesophyll cells with visible chloroplasts and nanoparticle aggregates within the cell wall matrix. b) 3D rendering of the same volume, with plant cells shown in green and nanoparticle aggregates highlighted in red.
The nanoholotomographic reconstructions showed that these particles were dispersed homogeneously throughout the leaf tissue and did not form aggregates larger than the spatial resolution of the technique. As a result, individual particles were not directly visible in the reconstructed volumes, indicating successful dispersion within the tissue. This behaviour was an explicit design objective of the nanoparticle formulation, as aggregation could impede transport or induce local toxicity. In this context, nanoholotomography serves not only as a localisation tool but also as a diagnostic method for assessing whether nanoparticle formulations behave as intended within living tissue.
Click image to enlarge
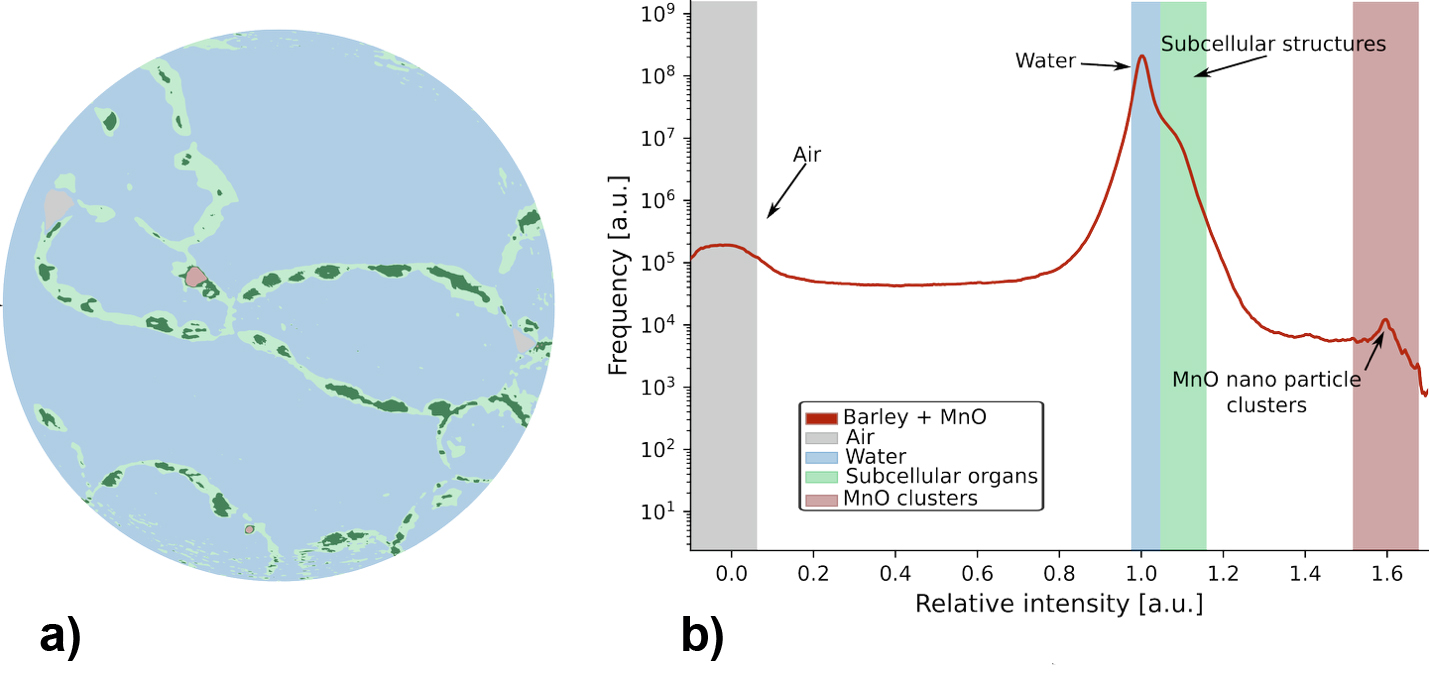
Fig. 2: Quantitative segmentation of living plant tissue containing nanoparticle aggregates. a) Segmented nanoholotomography slice distinguishing water and vacuoles (blue), cell walls (light green), chloroplasts (dark green), and nanoparticles (red). b) Histogram of relative pixel intensities corresponding to the mass densities of the different tissue components.
To capture nanoparticle localisation explicitly, aggregation was induced to generate clusters larger than the spatial resolution limit. In parallel experiments, mesoporous silica nanoshell (MSN) particles with stronger intrinsic aggregation tendencies were applied to tomato leaves. In this case, aggregates were directly visualised in three dimensions and shown to be located within cells immediately adjacent to the cell wall (Figure 2). Segmentation of the reconstructed volumes enabled quantitative differentiation between tissue components and nanoparticle clusters based on relative density contrasts.
The impact
The ability to image living plant tissue at the nanoscale represents a significant methodological advance. By eliminating the need for fixation, sectioning, or staining, nanoholotomography preserves native tissue architecture and enables direct observation of nanoparticle transport pathways. The visualisation of foliar-applied fertiliser nanoparticles within vascular and mesophyll tissues provides a foundation for understanding how such particles are loaded into transport networks and redistributed throughout the plant. These insights are essential for optimising foliar fertilisation strategies and, in the longer term, reducing fertiliser losses and their associated environmental impacts.
Principal publication
3D X-ray Microscopy Lights up Nanoparticles in Plants, E.V. Kristensen et al., ACS Nano, 19, 38910 (2025); https://doi.org/10.1021/acsnano.5c06203
References
[1] P.M. Kopittke et al., Environ. Int. 146, 106244 (2021).
[2] S. Husted et al., Trends Plant Sci. 28, 90 (2023).
| About the beamline: ID16B |
| ID16B is a hard X-ray nanoprobe designed for 2D and 3D analysis of heterogeneous materials, combining X-ray fluorescence (XRF), diffraction (XRD), absorption spectroscopy (XAS), excited optical luminescence (XEOL), X-ray beam-induced current (XBIC), and phase-contrast imaging. It supports low-temperature, in situ and operando sample environments. ID16B is dedicated to research areas of significant scientific and societal importance, including nanotechnology, Earth and environmental sciences, and biomedical research. |