- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2014
- Electronic structure and magnetism
- What drives oscillations in CO oxidation by O2 over Rh/Al2O3 catalysts?
What drives oscillations in CO oxidation by O2 over Rh/Al2O3 catalysts?
Spontaneous oscillatory behaviour in heterogeneous catalysis is of considerable interest from fundamental and applied points of view; anticipating and controlling such behaviour is important to process engineering, as it may be highly problematic to process control and efficiency.
Previous studies of oscillations in CO oxidation over Rh/Al2O3 systems have resulted in differing opinions as to their source. Shanks and Bailey [1] concluded that oscillations arose from temperature fluctuations within the reactor, whereas Ioannides et al. [2] preferred redox processes within the Rh phase as their preferred explanation, though no technique directly sensitive to the oxidation state of the Rh was applied.
To resolve this situation a 5wt%Rh/Al2O3 catalyst was investigated during CO oxidation catalysis using synchronous application of Rh K edge dispersive EXAFS (at beamline ID24), diffuse reflectance infrared spectroscopy (DRIFTS) and mass spectrometry (MS), at sub-second time resolution. [3]
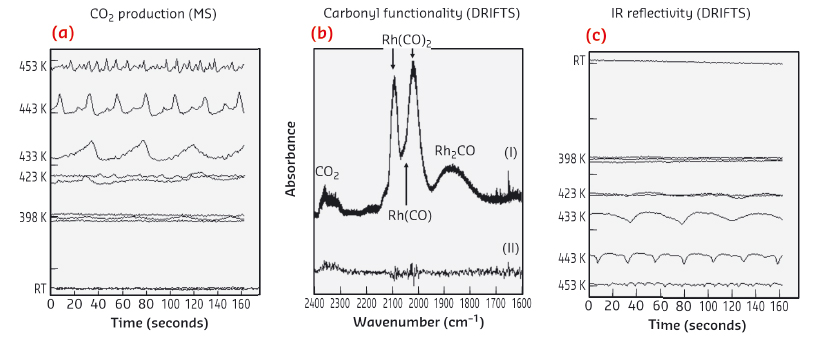
Figure 25a shows CO2 production at varying temperatures when a stoichiometric (2CO:O2) flow of reactive gas is passed over the Rh/Al2O3 catalyst. Rapid variations in CO2 evolution that vary in period/magnitude are observed. Figure 25b shows (I) DRIFTS during the oscillatory behavior at 433K and (II) the difference between the maximum and minimum levels of CO2 production. Linear, bridged and geminal carbonyls are observed but no relation to the oscillatory behaviour observed. These species do show that Rh is present in at least two forms, reduced nanoparticles (Rh(CO) and Rh2(CO)) and RhI(CO)2 centres, but cannot inform us as to whether redox (Rh(0) to Rh (3+)) bi-stabilities are directing these events.
 |
|
Fig. 25: a) Oscillations in CO2 (MS) as a function of temperature. b) DRIFTS showing (I) carbonyl speciation present during oscillatory behaviour at 433K, and (II) the difference in spectra taken at points of maximal and minimal levels of CO2 production. c) Temporal variation in overall infrared reflectivity. |
Figure 25c shows changes in infrared reflectivity (DRIFTS). Here we do observe variations that precisely match those observed in MS. These changes may have two origins: variations in surface temperature, or changes in the reflectivity of the Rh arising from a change from metallic nanoparticles to more oxidic entities. A precise correlation between these variations and the temperature of the sample can be established. However, this does not rule out variations (0 to +3) in the oxidation state of the Rh as the underlying source of the oscillations.
 |
|
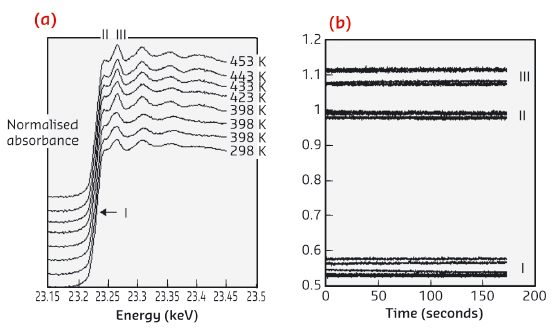
Fig. 26: a) Rh K edge XAFS at t = 0 seconds within the CO oxidation experiment as a function of temperature. b) Temporal variation observed at the three points indicated in (a) within the XAFS envelope. |
Figure 26 shows Rh K edge XAFS obtained at 0 seconds for each temperature. The XAFS envelope is different at each temperature reflecting changes in the balance between Rh in a metallic state and (RhI(CO)2). However, the temporal variation of the Rh K edge position (I), the Rh white line (II) – both very sensitive to the Rh0/Rh3+ ratio- and a second XANES feature (III) that indexes the Rh(0) nanoparticulate state (Figure 26b) again show no relationship to the oscillatory CO2 production.
As such, neither the carbonyl functionality (DRIFTS) nor the oxidation state of the Rh (XAFS) are the sources of the oscillations. Furthermore, the relationship between the temperature of the sample (Figure 25) and the production of CO2 shows that this cannot be the cause of the oscillations but rather a result of them. Therefore we have to search for another explanation that is invisible to both the DRIFTS and the Rh K edge EXAFS.
Previous studies [4] have shown that, alongside adsorbing molecular CO, in the regimes of Rh particle size and temperature investigated here, Rh nanoparticles also dissociate CO to yield adsorbed atomic carbon; a speciation invisible to infrared and Rh K edge XAFS. Also, the probability of CO dissociation as a function of temperature matches, in terms of onset and magnitude as a function of temperature, the oscillations in CO2 production we observe.
Therefore, the evidence points to a unique mechanism based upon CO dissociation. This leads to an accumulation and periodic ignition of adsorbed carbon as the source of the oscillatory phenomena.
Principal publication and authors
S.J.A. Figueroa (a,b) and M.A. Newton (a), J. Catal. 312, 69 (2014).
(a) ESRF
(b) Now at Brazilian Synchrotron Light Source (LNLS), Campinas (Brazil)
References
[1] B.H. Shanks and J.E. Bailey, J. Catal. 110, 197 (1988).
[2] T. Ioannides, A.M. Efstathiou, Z.L. Zhang and X.E. Verykios, J. Catal. 156, 265 (1995).
[3] M.A. Newton, Topics in Catalysis 52, 1410 (2009).
[4] V. Matolin, M.H. Elyakhloufi, K. Masek and E. Gillet, Catal. Letts. 21, 175 (1993).



