- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2013
- Structural biology
- Nucleotide specific bacterial mRNA ‘decapping’
Nucleotide specific bacterial mRNA ‘decapping’
The degradation of messenger RNA (mRNA) is an important means of controlling gene expression in all domains of life. The shorter the half-life of its mRNA, the more rapidly a cell can alter its gene expression programme in response to changes in the environment. While eukaryotic mRNAs are protected from ribonucleases by their 5’-methyl-guanosine cap structure, bacterial mRNAs are protected from degradation by the 5’-triphosphate group that remains on primary transcripts following transcription by RNA polymerase. Intriguingly, the enzymes that remove the cap structure from eukaryotic mRNAs (Dcp2) and the 5’-triphosphate from bacterial mRNAs (RppH) are members of the same ancient family of Nudix phosphohydrolases. In both cases, the resulting RNAs bear 5’ monophosphate groups that are targeted for degradation by 5’-3’ exoribonucleases or endoribonucleases with stimulatory 5’ monophosphate recognition domains.
A typical bacterial cell contains multiple Nudix hydrolases; Bacillus subtilis has 6, Escherichia coli has 13. It is not easy to tell from a simple homology search which of these proteins has RppH activity. A Blast search for homologs of B. subtilis RppH (BsRppH) in E. coli only identifies EcRppH in fifth position, for example. Biochemical verification is therefore currently required for enzyme identification. Structural studies of several of these enzymes might ultimately reveal sequence motifs important for distinguishing RppH proteins from other Nudix enzymes.
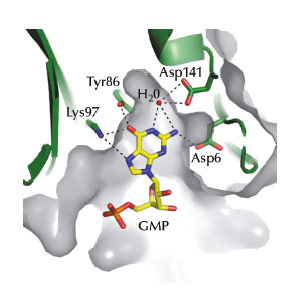
Using data collected at beamline ID14-1, we recently solved the crystal structure of RppH from the Gram-positive model organism B. subtilis bound to a two-nucleotide (nt) triphosphorylated RNA molecule (pppGpG). The structure suggested that, in addition to recognising the 5’ triphosphate group of the first residue of its substrates, BsRppH contains a pocket specific for guanosine (G) in the second position of bound RNAs (Figure 68). Indeed mutation of the guanosine in a known substrate of BsRppH to any other residue (A, C or U) reduced the activity of the enzyme by an order of magnitude. In addition to revealing the substrate specificity of BsRppH, the structure explained, through the relative positions of the magnesium ions in the catalytic site, why the B. subtilis enzyme removes the phosphate groups one at a time, while the E. coli and Bdellovibrio bacteriovorus enzymes release pyrophosphate (PPi) from their mRNA substrates.
 |
|
Fig. 68: The guanosine binding pocket of BsRppH. Hydrogen bonds between the protein and GMP are shown as dashed lines. |
The crystallography studies were complemented by NMR chemical shift mapping of BsRppH bound to one, two or three triphosphorylated nucleotides. The data showed that at least three nucleotides were required for optimal positioning of the RNA in the catalytic site. The structural studies were also supported by in vivo studies on a known substrate of BsRppH, the B. subtilis yhxA-glpP mRNA [1]. The second nucleotide of this mRNA is naturally a G-residue, consistent with the enzyme’s nucleotide preference in this position. In cells deleted for the rppH gene, the yhxA-glpP mRNA was stabilised about three-fold compared to wild-type cells. However, when the G in the second position was changed to U, this mRNA was stabilised regardless of the presence or absence of BsRppH in the cell, providing a nice confirmation of the importance of G2 in BsRppH substrates in vivo.
 |
|
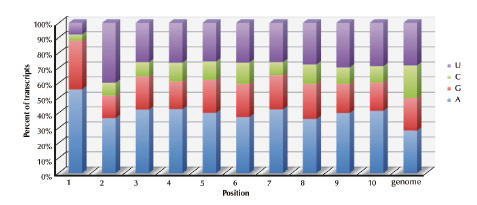
Fig. 69: Identity of the first 10 nucleotides in B. subtilis mRNA transcripts. Histogram showing the percent occurrence at each position of the nucleotides U (purple), C (green), G (red), A, (blue). The genome average is also shown. |
When one looks at primary transcripts in B. subtilis, it is clear that there is a counter-selection for mRNAs with G in the second position (Figure 69).
Only 15% of B. subtilis mRNAs contain a G2 residue, compared to 33% having G in position 1 (most RNA polymerases prefer to start with purine, A or G) and 21% in positions 3 to 10. This means that BsRppH can only act on a subset of B. subtilis mRNAs. A number of experiments have suggested that B. subtilis may have other as yet unidentified RNA pyrophosphohydrolases. It is anticipated that these enzymes will have a different substrate specificity to BsRppH.
Principal publication and authors
J. Piton (a), V. Larue (b), Y. Thillier (c), A. Dorléans (a), O. Pellegrini (a), I. Li de la Sierra-Gallay (a, d), J.J. Vasseur (c), F. Debart (c), C. Tisné (b) and C. Condon (a), Proc. Natl. Acad. Sci. USA 110, 8858-8863 (2013).
(a) CNRS UPR9073, Institut de Biologie Physico-Chimique, Paris (France)
(b) UMR 8015 CNRS/Université Paris Descartes, Paris (France)
(c) IBMM, UMR 5247 CNRS-Université Montpellier 1-Université Montpellier 2 (France)
(d) IBBMC, CNRS UMR8619, Université Paris-Sud, Orsay (France)
References
[1] P.-K. Hsieh, J. Richards, Q. Liu and J.G. Belasco, Proc. Natl. Acad. Sci. USA 110, 8864-8869 (2013).



