- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2010
- Structure of materials
- Flexible porous coordination polymers for selective adsorption
Flexible porous coordination polymers for selective adsorption
Among crystalline porous solids, metal organic frameworks (MOFs) deserve special interest. These hybrid solids, built up from inorganic subunits connected through polytopic ligands (such as poly-carboxylate and poly-pyrazolate) have the faculty to change their pore size reversibly upon external (temperature, pressure, light) or internal (entrapped guests) stimuli while remaining crystalline. This dynamic porosity leads to unusual sorption properties, which are attractive for applications in the area of fluid storage, capture or separation. One of the most archetypical examples of such a solid is MIL-53(M) (MIL stands for Materials Institute Lavoisier), a metal (M = Al, Cr, Fe) terephthalate built up from inorganic chains and linear ligands presenting one dimensional diamond-shaped micropores.
 |
|
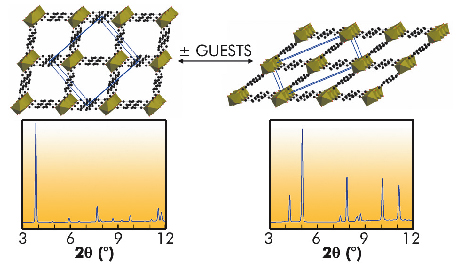
Fig. 52: The "breathing" behaviour of MIL-53 upon adsorption/desorption of guests, and the corresponding powder X-ray diffraction patterns which allow a direct evaluation of the pore size. Left: large-pore form; right: narrow-pore form. |
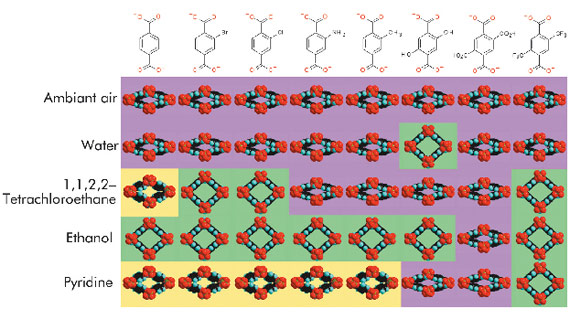
In the last few years, a multi-technique approach has been developed to study the adsorption of important gases (H2, CH4, CO2, alkanes, etc.) in these solids [1]. It could be of interest in industrial applications to obtain alcohol of very high purity. Therefore, the sorption of polar vapours (water, methanol and ethanol) in MIL-53(Cr) has been explored. Upon adsorption, the solid evolves from a large-pore form to a narrow-pore form (Figure 52), for which the pore volume depends on the nature of the guest. At higher alcohol pressure, the structure evolves once again to a large-pore form, whereas the narrow pore form is retained in the presence of water. The specific host-guest interactions associated with these adsorption processes were probed by microcalorimetry, X-ray structure investigation, IR and Raman spectroscopy, thermal analysis and molecular modelling. The in situ powder X-ray diffraction experiments were carried out at beamline BM01A. These studies revealed that the strength of the interaction depends on the nature of the adsorbed species (water < alcohols), and finally concludes that this solid is potentially interesting for the selective adsorption of alcohol from water/ethanol mixtures. Alternatively, the introduction of functional groups on the organic linker is an easy way to modify the pore surfaces, and thus to tune the sorption properties, affecting both the strength of the host-guest interactions and the pore opening. With this aim, various functionalised MIL-53(Fe) solids were prepared. While the hydrated form (under ambient air) of all these solids is associated with a narrow-pore form (see Figure 53), they present contrasting behaviours when immersed in liquids.
Powder X-ray diffraction indeed reveals that the pore opening (and thus the number of adsorbed molecules), strongly depends on the nature of both the liquid and the grafted group (Figure 53). Besides the appearance of a large-pore form requires that the host-guest interactions (hydrogen bonding) overcome the intraframework interactions. Providing that this requirement is fulfilled, the adequacy of the chemical nature of both components (liquids-grafted groups) could lead to selective adsorption.
Principal publications and authors
S. Bourrelly (a), B. Moulin (b), A. Rivera (c), G. Maurin (c), S. Devautour-Vinot (c), C. Serre (d), T. Devic (d), P. Horcajada (d), A. Vimont (b), G. Clet (b), M. Daturi (b), J.-C. Lavalley (b), S. Loera-Serna (a), R. Denoyel (a), P.L. Llewellyn (a) and G. Férey (d), J. Am. Chem. Soc. 132, 9488-9498 (2010); T. Devic (d), P. Horcajada (d), C. Serre (d), F. Salles (c), G. Maurin (c), B. Moulin (b), D. Heurtaux (d), G. Clet (b), A. Vimont (b), J.-M. Grenèche (e), B. LeOuay (d), F. Moreau (d), E. Magnier (d), Y. Filinchuk (f), J. Marrot (d), J.-C. Lavalley (b), M. Daturi (b) and G. Férey (d), J. Am. Chem. Soc. 132, 1127-1136 (2010).
(a) Laboratoire Chimie Provence, CNRS, Université Aix-Marseille (France)
(b) Laboratoire Catalyse et Spectrochimie, CNRS, ENSICAEN, Université de Caen (France)
(c) Institut Gerhardt, CNRS, ENSCM, Université de Montpellier (France)
(d) Institut Lavoisier de Versailles, CNRS, Université de Versailles St-Quentin en Yvelines (France)
(e) Laboratoire de Physique de l'Etat Condensée, CNRS, Université du Maine (France)
(f) Swiss Norwegian Beamline, ESRF, Grenoble (France)
References
[1] C. Serre, S. Bourrelly, A. Vimont, N.A. Ramsahye, G. Maurin, P.L. Llewellyn, M. Daturi, Y. Filinchuk, O. Leynaud, P. Barnes and G. Férey, Adv. Mater. 19, 2246-2251 (2007).