- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2009
- Structural biology
- Abundance of cofactors in photosystem II
Abundance of cofactors in photosystem II
Oxygenic photosynthesis is driven by photosystems I and II (PSI and PSII), two large protein-cofactor complexes found in the thylakoid membranes of plants, algae and cyanobacteria. The structure of PSII is of special interest as it contains the unique Mn4CaOx cluster that catalyses the photolysis of water: 2H2O ![]() O2 + 4H+ + 4e-. These electrons reduce plastoquinone (PQ) at the QB-site to plastoquinol (PQH2) with the concomitant uptake of protons from the cytosol:
O2 + 4H+ + 4e-. These electrons reduce plastoquinone (PQ) at the QB-site to plastoquinol (PQH2) with the concomitant uptake of protons from the cytosol:
![]()
 |
|
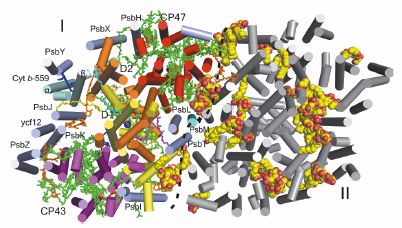
Fig. 108: The homodimeric PSII complex. Overview of PSII from the cytoplasmic side (membrane-extrinsic subunits omitted). The monomer-monomer interface is indicated by a black dashed line. Helices are shown as cylinders with the subunits D1 (yellow), D2 (orange), CP43 (magenta), CP47 (red), cyt b-559 (cyan, subunits |
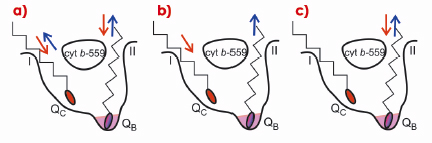
The crystal structure of homodimeric PSII at 2.9 Å resolution was determined from data collected at beamline ID14-2. Each monomer (see Figure 108) contains 20 assigned protein subunits and 92 cofactors (35 chlorophylls, two pheophytins, two haems, 12 ß-carotenoids, 25 lipids, seven detergent molecules (ß-dodecylmaltoside), three plastoquinones, the Mn4Ca cluster, one bicarbonate, two Ca2+, one Fe2+, and one Cl-). The surprisingly high amount of lipids in comparison with other complexes involved in oxygenic photosynthesis might be related to special functions of lipids in PSII. Lipids found at the monomer-monomer interface and around the reaction centre seem to be an ideal lubricating component involved in the processes of dis- and reassembly of PSII needed for the replacement of photodamaged subunit D1 [1]. The largest lipid cluster, composed of eight lipids, forms an intrinsic lipid bilayer providing a hydrophobic environment in the large plastoquinone/plastoquinol exchange cavity that features two channels connecting the binding site of plastoquinone QB with the plastoquinone pool in the thylakoid membrane. A newly observed third plastoquinone (QC), in combination with the positions of the channels, allowed us to propose possible mechanisms for rapid plastoquinone/plastoquinol exchange in PSII (Figure 109): a) alternating mechanism, where both channels work as entry/exit in an alternating way; b) wriggling, where one channel is exclusively for entry and the other is only for exit; c) single channel mechanism, where only one channel is involved in plastoquinone/plastoquinol exchange.
 |
|
Fig. 109: Simplified scheme of plastoquinone/plastoquinol exchange in PSII. a) Alternative mechanism, both channels work as entry/exit; b) wriggling – channel I is only for entry and channel II for exit; c) single channel mechanism – only channel II is used for exchange. Red arrows indicate movement of PQ, blue of PQH2. See principal publication for details. |
Additionally, four smaller lipid clusters within the PSII monomer may participate in auxiliary oxygen channelling to the cytosolic side of the thylakoid membrane. This is shown by the positions of xenon atoms in the crystal structure of PSII derivatised with gaseous xenon (part of the data collected at beamlines ID23-1 and ID23-2) that might indicate pathways for oxygen release from the Mn4CaOx cluster.
A combination of native and anomalous data (collected at ID29) allowed us to assign unambiguously a chloride ion (as well as its analogue bromide) in close vicinity to the Mn4CaOx cluster. The chloride ion is located 6.5 Å from the cluster in a positively charged environment provided by strictly conserved amino acids and connected with the cluster via putative water molecule(s). A possible shift in the position of Mn ions due to radiation damage (reduction of Mn(III) and Mn(IV) to Mn(II)) must be kept in mind.
Theoretical calculations using the obtained structural model allowed us to describe a complex transport system within PSII. The arrangement of possible substrate and product channels suggests a strict spatial separation of water, oxygen and proton fluxes to and from the Mn4CaOx cluster. We identified nine distinct channels connecting the cluster with the lumenal side of PSII, the four wider ones and the five narrower ones being possible water and oxygen or proton channels, respectively. The position of chloride in the proton channel(s) might be necessary to facilitate proton escape from the cluster by tuning pKa values of the surrounding amino acids and excludes its direct involvement in the water splitting chemistry. The nature of the oxygen channels was recently confirmed by derivatisation of PSII crystals with gaseous krypton and co-crystallisation with DMSO [2].
Acknowledgements
This work was supported by grants from the German Research Council (SFB 498, projects A4, C7).
References
[1] Y. Nishiyama, S.I. Allakhverdiev and N. Murata, Biochim Biophys Acta 1757, 742 (2006).
[2] A. Gabdulkhakov, A. Guskov, M. Broser, J. Kern, F. Müh, W. Saenger and A. Zouni, Structure 17, 1223 (2009).
Principal publication and authors
A. Guskov (a), J. Kern (b, c), A. Gabdulkhakov (a), M. Broser (b), A. Zouni (b) and W. Saenger (a), Nat. Struct. Mol. Biol. 16, 334 (2009).
(a) Institut für Chemie und Biochemie/Kristallographie, Freie Universität Berlin (Germany)
(b) Institut für Chemie/Max Volmer Laboratorium für Biophysikalische Chemie, Technische Universität Berlin (Germany)
(c) Physical Biosciences Division, Lawrence Berkeley National Laboratory, Berkeley (USA)



