- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2004
- Soft Condensed Matter
- Structure of Chocolate Clarified with Synchrotron Powder Diffraction Data
Structure of Chocolate Clarified with Synchrotron Powder Diffraction Data
Most chocolate eaters will have had the surprising experience that a newly opened bar of chocolate (pure or milk) has a greyish-white layer instead of the familiar chocolate colour (Figure 61). Most likely, this bar has fallen victim to the development of fat bloom. The cocoa butter in chocolate is responsible for chocolate's appearance. In most consumer chocolate the cocoa butter is in its second highest melting form, called ß(V). Storage of ß(V) chocolate at too high temperatures induces the transition to the most stable ß(VI) form and this transition is commonly associated with fat bloom. To understand the ß(V) ![]() ß(VI) transition, crystal-structure knowledge of the principal components of cocoa butter is required. At room temperature solid cocoa butter consists mainly of three mono-unsaturated triglycerides, 1,3-dipalmitoyl-2-oleoylglycerol (POP), 2-oleoyl-1-palmitoyl-3-stearoylglycerol (POS) and 1,3-distearoyl-2-oleoylglycerol (SOS). The latter triglyceride comprises ~25% of cocoa butter and is known to play a major role in the crystallisation of cocoa butter into the ß forms. Good quality single crystals of SOS are extremely difficult to obtain so we resorted to synchrotron powder diffraction as an alternative.
ß(VI) transition, crystal-structure knowledge of the principal components of cocoa butter is required. At room temperature solid cocoa butter consists mainly of three mono-unsaturated triglycerides, 1,3-dipalmitoyl-2-oleoylglycerol (POP), 2-oleoyl-1-palmitoyl-3-stearoylglycerol (POS) and 1,3-distearoyl-2-oleoylglycerol (SOS). The latter triglyceride comprises ~25% of cocoa butter and is known to play a major role in the crystallisation of cocoa butter into the ß forms. Good quality single crystals of SOS are extremely difficult to obtain so we resorted to synchrotron powder diffraction as an alternative.
 |
Fig. 61: a) Chocolate, b) Chocolate affected by fat bloom. |
High-resolution powder diffraction data for ß2-SOS, the second highest melting form of SOS, were collected at BM16 and for cocoa butter from the Ivory Coast in the ß(V) phase at the Swiss-Norwegian CRG BM01B. Interestingly, both diffraction patterns are very similar, suggesting they share the same principal structural features. Unit-cell determination ('indexing') was a bottleneck for a long time but by developing our own indexing program this problem was overcome. In retrospect, the indexing problems were caused by the combination of a triclinic unit cell and exceptionally large differences of the cell-axis lengths, the shortest being ~5.45 Å, the longest ~ 65 Å. Also the pattern of ß(V) cocoa butter could be indexed, which is surprising because cocoa butter is a co-crystallised mixture of triglycerides. For crystal structure determination of ß2-SOS we used the direct-space programs FOX and ORGANA that are both based on R-value minimisation and exclusion of chemically unfeasible types of packing. Starting from the SOS structural model, a crystal structure of ß(V) cocoa butter was found in a similar way, with the outer ethyl-groups in the saturated chains being partially occupied.
Both ß2-SOS and ß(V) cocoa butter have a triple-chain length packing with alternating layers of saturated chains (being packed together) and mono-unsaturated oleoyl chains. With respect to the oleoyl layer, the crystal structures of ß2-SOS and ß(V) cocoa butter differ considerably from the packing model for SOS proposed in the literature because of the absence of - interaction (Figure 62). This latter feature and the cocoa butter crystal structure model provide an explanation for the driving force behind the slow ß(V) ![]() ß(VI) phase transition. The packing of SOS, POS and POP in ß(V)-CB has a ß2-SOS like structure in which on average only 57% of the two carbon positions at the end of the saturated chains are occupied, while 43% are ethyl-end 'holes'. The absence of interactions at these 'holes', and the absence of - interaction in the oleoyl layer, causes ß(V) to be metastable and is likely to be the driving force for the transformation to the ß(VI) form, implying a structural re-organisation that reduces the amount of directly opposing 'holes'. It can be envisaged that this process may take place either via a solid-state ß(V) to ß(VI) transformation or via a melt-mediated process in which the ß(V) dissolves in the melt and ß(VI) crystallises. This fundamental knowledge concerning the stability of the cocoa butter ß phases has already led to the development of a new chocolate-manufacturing process technology in which crystallisation of chocolate into the ß(V) and also into the ß(VI) can be regulated by using the appropriate liquid seed crystals and temperature handling[1].
ß(VI) phase transition. The packing of SOS, POS and POP in ß(V)-CB has a ß2-SOS like structure in which on average only 57% of the two carbon positions at the end of the saturated chains are occupied, while 43% are ethyl-end 'holes'. The absence of interactions at these 'holes', and the absence of - interaction in the oleoyl layer, causes ß(V) to be metastable and is likely to be the driving force for the transformation to the ß(VI) form, implying a structural re-organisation that reduces the amount of directly opposing 'holes'. It can be envisaged that this process may take place either via a solid-state ß(V) to ß(VI) transformation or via a melt-mediated process in which the ß(V) dissolves in the melt and ß(VI) crystallises. This fundamental knowledge concerning the stability of the cocoa butter ß phases has already led to the development of a new chocolate-manufacturing process technology in which crystallisation of chocolate into the ß(V) and also into the ß(VI) can be regulated by using the appropriate liquid seed crystals and temperature handling[1].
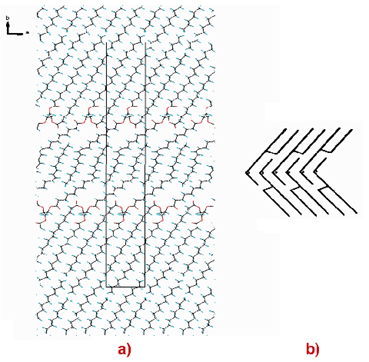 |
Fig. 62: a) Crystal structure packing and characteristics of ß2-SOS and ß(V)-cocoa butter. b) Packing of SOS proposed in the literature. |
In conclusion, on the basis of highresolution synchrotron powder data we solved the crystal structures of ß2-SOS and ß(V) cocoa butter, and thus obtained the crystal structure of the most commonly produced form of chocolate. The packing of the crystal structure models provides an explanation of their metastability and it is expected that this will form the basis of an atomic-level description of the ß(V) ![]() ß(VI) phase transition, which is associated with the occurrence of fat bloom.
ß(VI) phase transition, which is associated with the occurrence of fat bloom.
References
[1] K.F. Van Malssen, A.J. Van Langevelde, R. Peschar, H. Schenk. Patent WO 01/06863 (2001).
Principal Publications and Authors
R. Peschar, M.M. Pop, D.J.A. De Ridder, J.B. Van Mechelen, R.A.J. Driessen, H. Schenk, J. Phys. Chem. B. 108(40), 15450-15453 (2004).
Laboratory for Crystallography, HIMS, University of Amsterdam (The Netherlands)



