- Home
- News
- Spotlight on Science
- Spatially resolving...
Spatially resolving the state of charge in Li-ion electrodes
18-07-2019
Quantifying heterogeneities in Li-ion electrodes is crucial for understanding degradation mechanisms. Using high-speed and high-resolution XRD and XRD-CT, charge balancing kinetics due to sub-electrode potential differences were quantified, as well as and inter and intra particle lithiation heterogeneities.
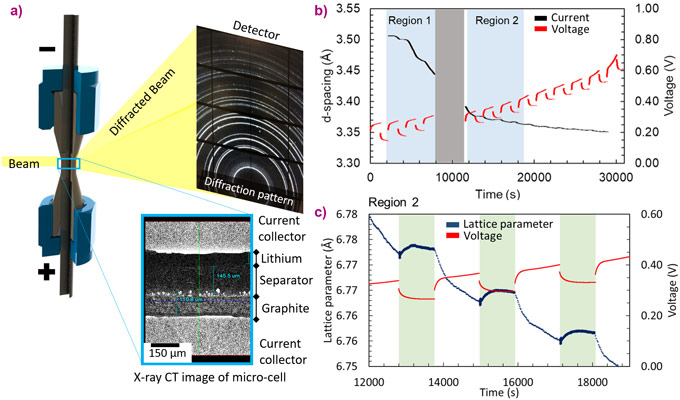
High-speed X-ray diffraction (XRD) and region-of-interest XRD-computed tomography (ROI XRD-CT) have been used in concert to monitor charge-balancing kinetics and spatial heterogeneities of charge in composite silicon (Si)-graphite electrodes of a Li-ion battery. As an electrode material, Si holds tremendous potential for use in high energy density Li-ion cells. Most Li-ion cells use graphite as a negative electrode, but Si has a theoretical capacity of >10× that of graphite. The drawback of high Si content electrodes is that, currently, they suffer from severe degradation. Consequently, in state-of-the-art high-energy density batteries where cycle life is important, Si is typically only utilised in combination with graphite but only in small amounts (< 10% mass). Graphite and Si have different electrochemical and mechanical properties, which results in the two materials having different responses when subject to operating conditions inside a Li-ion cell [1]. For example, under certain operating conditions one material might change its potential difference (vs. Li) faster than the other. This causes charge imbalances that equilibrate when the load on the Li-ion cell is removed, in a process known as relaxation [2]. The charge balancing kinetics, as well as the spatial distribution of charge inside composite electrodes is not well understood but plays a key role in the performance and degradation of the electrode.
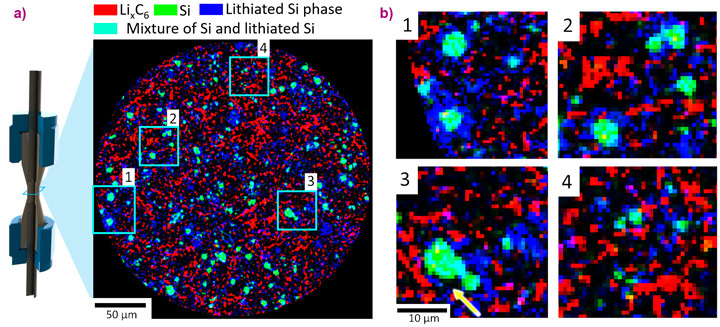
At beamline ID15A, high-speed (up to 100 Hz) and high energy (50 keV) XRD was used to monitor the state of charge of the graphite phase during operation and the moments following a halt in operation when relaxation kinetics occurs (Figure 1). The graphite phase was shown to continue to change its state of charge following a halt in operation which indicated that the Si and graphite phases continued to have electrochemical activity, where lithium from one material was transported to the other due the potential difference that formed between the two materials during operation. ROI XRD-CT with a resolution of 1 µm facilitated mapping out the distribution of lithiation between the graphite and Si phases both before and after the delithiation process. The graphite phase displayed significant heterogeneity in lattice parameter, indicating that the potential difference between the different lithiation extents was not high enough to induce charge equilibration within the graphite portions of the electrode. When Si particles lithiate, they form a shell of lithiated amorphous phases on top of a crystalline Si core. This core-shell structure of the Si particles was observed, where the shell phase was partially lithiatied Si and the core phase is non-lithiated crystalline Si (Figure 2). The tomographically reconstructed core and shell diffraction patterns were analysed, revealing that the shell phase was semi-crystalline indicating sub-micrometre heterogeneities. The presence of crystalline Si within the core showed that lithiation equilibration did not occur inside the Si particles, which was most likely due to internal strains hindering the lithiation reaction front. The presence and extent of the remaining crystalline Si showed that the electrode was significantly underutilised.
The cutting-edge capabilities of beamline ID15A facilitated spatially and temporally resolved crystallographic information from operating Li-ion electrodes. Inter and intra particle heterogeneities were quantified in 3D and over time, demonstrating high-speed XRD and ROI XRD-CT as a powerful diagnostic couple for understanding performance limitations and degradation mechanisms of Li-ion electrodes. This opens a plethora of opportunities for spatial crystallographic investigations of next generation battery electrodes, some of which are plagued by mechanical degradation stemming from sub-particle crystallographic defects and heterogeneities. These details can now be spatially and temporally resolved providing a valuable tool to design and optimise next generation battery materials.
Principal publication and authors
Spatially resolving lithiation in silicon-graphite composite electrodes via in situ high-energy X-ray diffraction computed tomography, D.P. Finegan (a), A. Vamvakeros (b,c), L. Cao (a), C. Tan (d,e), T.M.M. Heenan (d,e), S.R. Daemi (d), S.D.M. Jacques (c), A.M. Beale (c,f,g), M. Di Michiel (b), K. Smith (a), D. J.L. Brett (d,e), P.R. Shearing (d,e), C. Ban (a), Nano Lett., 19, 3811-3820 (2019), doi: acs.nanolett.9b00955.
(a) National Renewable Energy Laboratory, Golden (USA)
(b) ESRF
(c) Finden Limited, Abingdon (UK)
(d) Electrochemical Innovation Laboratory, Department of Chemical Engineering, University College London (UK)
(e) The Faraday Institution, Harwell Science and Innovation Campus, Didcot (UK)
(f) Department of Chemistry, University College London (UK)
(g) Research Complex at Harwell, Harwell Science and Innovation Campus, Rutherford Appleton Laboratories, Didcot (UK)
References
[1] K.P. Yao, et al., Operando quantification of (de)lithiation behavior of silicon-graphite blended electrodes for lithium-ion batteries, Advanced Energy Materials, 9, 1803380 (2019).
[2] H. Tanida, et al., Elucidating the driving force of relaxation of reaction distribution in LiCoO2 and LiFePO4 electrodes using X-ray absorption spectroscopy, J. Phys. Chem. C, 120, 4739 (2016).