- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2010
- Electronic structure and magnetism
- Photomagnetic molecular magnet
Photomagnetic molecular magnet
One of the current challenges in magnetism is the design of nanometric magnetic materials, such as thin films, nanoparticles and high spin molecules. The advantage of developing molecular magnets is their well-defined sizes and the flexibility of the constituent metal ions. During the last decade, the field of molecular magnetism has been driven by tailoring molecular magnets that could be activated by external parameters such as temperature, light irradiation or pressure [1]. The family of Prussian blue analogues is of particular interest with the recently synthesised copper octacyanomolybdate presenting interesting photomagnetic properties. Indeed the molecular system goes from a non-magnetic (paramagnetic behaviour) to a magnetic state. The molecules undergo a reversible magnetic transition under light irradiation for which the origin remains puzzling.
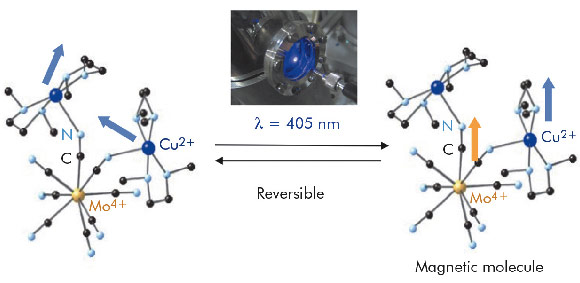
We will focus on [Mo(CN)6(CN-CuL)2], L being N,N',dimethyl ethylene diamine (labelled MoCu2-Meen). Before irradiation, the molecules behave as uncoupled CuII paramagnetic ions (with central S=0 diamagnetic MoIV ion) whereas after irradiation (405 nm) they behave as superparamagnetic molecules with one CuII ions coupled to the molybdenum ion (see Figure 76). The commonly proposed mechanism based on classical SQUID measurements is a photoinduced charge transfer from MoIV (S = 0) to CuII (S = 1/2) leading to the formation of MoV (S = 1/2) and CuI (S = 0) with strong ferromagnetic coupling between the other CuII spin carriers.
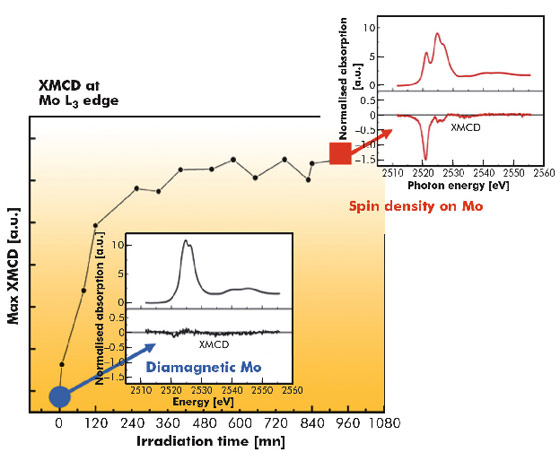
X-ray magnetic circular dichroim (XMCD) spectroscopy provides a unique tool to identify the magnetic states of the different metallic ions. In this highlight we present Mo L2,3 edge measurements that probe the 4d electronic occupation and magnetic state. XMCD gives the oxidation state and the spin state of the molybdenum ion. Beamline ID12 is unique in that it can provide circular polarisation at Mo L2,3 edges. We used its EMPHU undulator that emits flexible polarisation (circular left and circular right). At this energy range (2500-2610 eV) the polarisation rate delivered by the Si(111) double crystal monochromator is very low: it drops down to about 12% at the Mo L3 edge (2523 eV) and 4% at the Mo L2 edge (2629 eV). The difficulty of the experiment resides also in the particular low X-ray flux configuration that we adopted to avoid radiation damage: ESRF 4-bunch mode and 25-µm thick aluminium foil inserted upstream.
X-ray and laser (405 nm) induced photomagnetic phases were both studied by XMCD and found to be essentially identical. We measured the XMCD spectrum in a magnetic field of ± 6 T and at 10 K before and after photoconversion.
 |
|
Fig. 76: Photomagnetic transition schema of the MoCu2-Meen molecule. Photoconversion occurs with a laser (405 nm) installed in front of the viewport (see the image) or directly by the X-ray photons. |
Figure 77 shows the XMCD signal before and after irradiation. Before irradiation, no signal is found as expected for the spin zero MoIV ion. After irradiation, the measured XMCD signal shows the molybdenum magnetic transformation. The mainly negative XMCD signal shows that the Mo spin is in the direction of the magnetic field. After careful normalisation of the spectra, we extracted the oxidation state and magnetic spin moment of molybdenum. We found that the molybdenum photomagnetic state is S = 1 high spin MoIV: the two electrons of MoIV (4d2) are in two different non degenerate 4d orbitals and gives an S = 1 spin. There is no change in the oxidation state of the Mo ions and the only way to explain the existence of spin density evidenced by the XMCD signal is to suppose the formation of high spin MoIV (S = 1) entities. Under such a hypothesis, the X-ray photo-induced species would be Mo(HS)IVCuII2–Meen (HS = high spin). This new experimental finding has been supported by a very recent theoretical model developed by Carvajal et al. [2].
Our XMCD measurements have established the presence of an unsuspected high spin MoIV species that has never been observed before. This might explain highly energetic photomagnetic properties and pave the way towards the understanding of molybdenum based photomagnetic compounds. Such a magnetic characterisation could only be done by XMCD measurements.
Principal publication and authors
M-A. Arrio (a), J. Long (b), C. Cartier dit Moulin (b), A. Bachschmidt (b), V. Marvaud (b), A. Rogalev (c), C. Mathonière (d), F. Wilhelm (c) and Ph. Sainctavit (a), J. Chem. Phys C. 114, 593–600 (2010).
(a) Institut de Minéralogie et de Physique des Milieux Condensés, CNRS/UPMC/IPG/IRD, (France)
(b) Institut Parisien de Chimie Moléculaire, CNRS/UPMC (France)
(c) ESRF
(d) Institut de la Matière Condensée de Bordeaux, CNRS/Université Bordeaux 1 (France)
References
[1] O. Sato, J. Photochem. Photobio. C: Photochem. Rev. 5, 203-223 (2004).
[2] M.-A. Carvajal, M. Reguero and C. de Graaf, Chem Com. 46, 5737-57399 (2010).