- Home
- News
- Spotlight on Science
- New structures of...
New structures of carbonates at high pressures and their relevance for the deep Earth carbon cycle
19-02-2013
Calcite and dolomite, two of the most common minerals on the Earth’s surface, undergo phase transitions at high pressure. Their structures have be identified by single-crystal diffraction experiments under extreme conditions. Dolomite-III, in particular, was found to be triclinic with 80 atoms in the unit cell and has unexpected structural complexity. It is thermodynamically stable at the high pressures and temperatures found in the Earth’s interior, and may constitute a significant carbon repository in the lower mantle.
Calcite, CaCO3, and dolomite, CaMg(CO3)2 are two very common minerals on the Earth’s surface. They are probably formed in the deep sea sediments and coral reefs. The chemical equilibria regulating their precipitation is affected by the CO2 content in the oceans and atmosphere, two parameters which are directly related to the Earth’s actual and past climatic conditions. During rock-forming processes, sedimentary rocks containing carbonate-bearing sediment are transformed into limestone. These rocks can be observed in mountainous regions, for example they are the main outcrops in the area surrounding Grenoble. Their study may therefore provide fundamental insight into the evolution of the climate throughout the Earth’s history. However, not all of the limestone gets tranformed into mountains as a significant portion of the marine carbonates are transferred into the deep Earth through the subduction mechanism of the oceanic crust and its overlying sediment layers. The transformation of carbonates into the Earth’s mantle may lead to the storage of carbon at depth, whereas the return of carbon to the Earth’s surface may occur through volcanic processes. Detailed knowledge of the fate of carbonates in the Earth’s mantle is important for a precise estimation of the carbon content of our planet and to build accurate models for the description of the global carbon cycle and the evolution of the Earth’s climate. The first step in any of these investigations, however, is to assess the structures of these carbon-rich materials at extreme conditions.
We focussed in particular on the phase behaviour of dolomite in order to study the phase evolution at the high values of pressure and temperature expected in the Earth’s mantle. Very tiny single crystal samples were pressurised in diamond anvil cells up to a pressure of 80 GPa, and the structure evolution of dolomite was monitored by X-ray diffraction. The experiments were performed at the beamline ID09A, employing the recently optimised single-crystal diffraction setup, well-suited to the investigation of samples of a few µm3 in size at pressure values in excess of a megabar. With increasing pressure, dolomite undergoes two structural transitions above 17 GPa and 35 GPa respectively. The first one is a displacive transition towards a lower symmetry structure (dolomite-II), very similar to the one encountered in CaCO3 at much lower pressures [1]. The second transition (dolomite-III) exhibits a first-order character. Despite the significant volume reduction (5%) during the transition from dolomite-II to dolomite-III, a single crystal domain with a quality suitable for structural investigation was preserved (Figure 1).
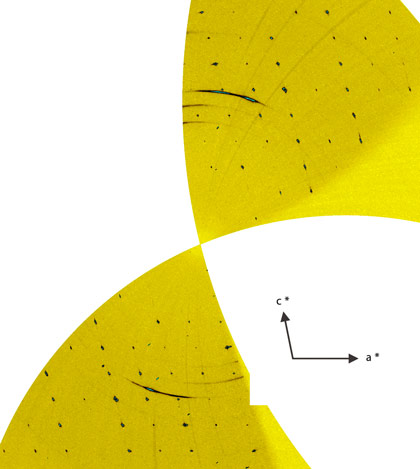 |
|
Figure 1. Experimental reconstructed h0l reciprocal space of dolomite-III with diffraction data collected at 55 GPa. |
Dolomite-III is triclinic, with 80 atoms in the unit cell (Figure 2). The main structural feature is the presence of non-coplanar CO3 groups, a feature also present in the recently solved CaCO3-III structure. Moreover, the presence of a further chemically bonded oxygen close to the carbon positions indicates that this structure represents an intermediate topology between the low pressure carbonates with planar CO3 groups and the ultrahigh pressure carbonates, with tetrahedral coordinated carbon, which have been predicted but not yet observed experimentally [2].
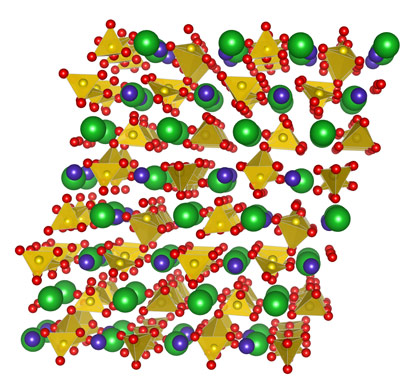 |
|
Figure 2. Crystal structure of dolomite-III at 55 GPa (Green: Ca; blue: Fe; red: O; yellow: C). |
To conclude, the thermodynamic stability of dolomite-III at combined ultrahigh pressures and temperatures was investigated using a recently developed laser heating system for single crystal diffraction. Our results indicate that single-phase dolomite-III is stable at the high pressures and temperatures existing in the lower mantle, in agreement with a recent powder diffraction study [3]. Dolomite-III may therefore act as an important carbon repository in the lowermost mantle. The similarity between dolomite-III and calcite-III even suggests the possibility of an extended solid-solution between CaCO3 and CaMg(CO3)2, with this new structure acting therefore as the main vehicle for carbon transport in subduction environments; that is, from the crustal environments towards the inner Earth.
Principal publication and authors
Structures of dolomite at ultrahigh pressure and their influence on the deep carbon cycle, M. Merlini (a), W.A. Crichton (b), M. Hanfland (b), M. Gemmi (c), H. Müller (b), I. Kupenko (d), L. Dubrovinsky (d), Proc. Natl. Acad. Sci. U.S.A. 109, 13509-13514 (2012).
(a) Dipartimento di Scienze della Terra, Università degli Studi di Milano (Italy)
(b) ESRF
(c) Center for Nanotechnology Innovation@NEST, Istituto Italiano di Tecnologia, Pisa (Italy)
(d) Bayerisches Geoinstitut, Universität Bayreuth (Germany)
References
[1] L. Merrill and W.A. Bassett, Acta Cryst. B31, 343–349 (1975).
[2] A. Oganov et al., Earth Planet. Sci. Lett. 273, 38–47 (2008).
[3] Z. Mao et al., Geophys Res. Lett. 38, L22303 (2011).
Top image: The dolomite sample after high-pressure and high-temperature experiments up to melting point.



