- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2017
- Matter at extremes
- Ionic ice mixture under pressure
Ionic ice mixture under pressure
Knowing the high-pressure behaviour of water-ammonia mixtures has strong relevance for the description of giant icy planets. Here, it is demonstrated that under pressure, the equimolar mixture transforms into an unusual solid alloy composed of both molecular and ionic species, owing to a topological frustration.
Water and ammonia are simple molecules, abundant in the universe. Together with methane, they are thought to be the main constituents of the internal layers of the giant icy planets Uranus and Neptune, where extreme pressure-temperature conditions exist. It is suspected that the unusual magnetic field of these planets originates from the ionisation of such molecules at high pressures and temperatures. Previous studies have shown that for the pure ices, pressure favours the creation of ions at pressures in excess of 100 GPa at 300 K, but the presence of ionic species in ice mixtures had not been experimentally evidenced.
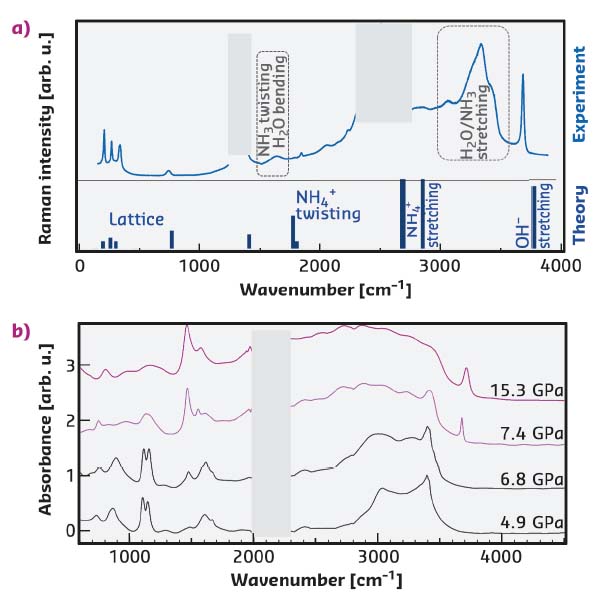
Experiments and first-principles simulations were used to study the solid phases of an equimolar mixture of water and ammonia, known as ammonia monohydrate (AMH). Several polymorphs had previously been disclosed below 10 GPa, all described as hydrogen-bonded molecular solids. Using Raman and synchrotron IR spectroscopies, hydroxyl (OH–) and ammonium (NH4+) ion signatures were detected in the AMH samples compressed above 7.4 GPa (Figure 48). These spectral bands occur at frequencies close to those expected for a recently predicted P4/nmm structure. However, this structure is fully ionic, i.e. solely composed of hydroxyl and ammonium ions, while the presence of strong Raman and IR bands from H2O and NH3 molecules indicate only partial ionisation of AMH. X-ray diffraction experiments conducted at ID27 revealed the sample was composed of two phases: a body-centred cubic (bcc) phase, representing the major part, and the P4/nmm phase. The bcc phase had been previously observed in neutron diffraction experiments and described as a disordered molecular alloy (DMA) where H2O and NH3 molecules are randomly distributed on the bcc lattice. The coexistence of a purely molecular alloy with a purely ionic phase was highly intriguing. Computer simulations of the bcc phase were conducted to better understand its nature.
 |
|
Fig. 48: a) Raman spectra of AMH at 10 GPa. The experimental spectra (collected at RT) and the theoretical one (calculated at 0 K) for the fully ionic P4/nmm structure are shown in the upper and lower panels, respectively. b) Experimental IR absorption spectra of AMH collected upon decompression at RT at SOLEIL synchrotron. Pink and black curves are spectra above and below the ionic-molecular phase transition, respectively. |
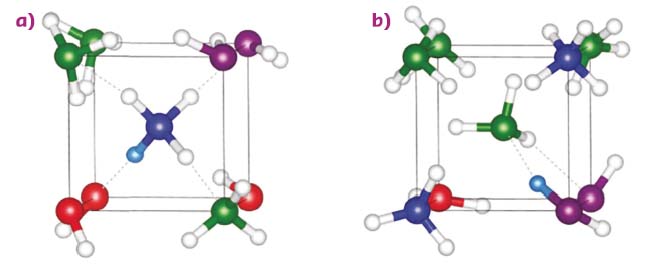
The results showed that the bcc phase is partly ionic, containing up to 36 % NH4+·OH– pairs at 10 GPa at 300 K, due to a proton transfer from a H2O to a NH3 molecule (Figure 49a). The OH– and NH4+ vibrations occur at nearly the same frequencies as in the P4/nmm crystal, making it hard to distinguish the respective contributions of the two phases in the vibrational spectra. Part of the molecules does not ionise in the bcc phase (baptised disordered ionic-molecular alloy, or DIMA) due to a topological frustration: the substitutional disorder of the bcc structure implies that two NH3 molecules can be first-neighbours, and if one of those molecules receives a proton from nearby water, then Coulombic repulsion hinders the formation of another cation in its first-neighbour shell (Figure 49b). Calculations also show that the DIMA phase has a higher energy than P4/nmm, indicating that the system is kinetically trapped in the DIMA phase at 300 K.
 |
|
Fig. 49: Simulation snapshots showing the nearest-neighbour environment of a) an ammonium ion or b) an ammonia molecule. The H atom involved in the H-bond between the central ammonia and neighbour water molecule is rendered in cyan, the others are represented by white spheres. N atoms are in green and blue for NH3 and NH4+, respectively, and O atoms are in magenta and red for H2O and OH–, respectively. Solid and dashed lines depict the cubic unit cell and the H-bonds, respectively. |
The present study demonstrates that AMH spontaneously converts into an unusual crystalline state above 7.4 GPa, where H2O and NH3 molecules coexist with OH– and NH4+ ions. The relatively low-pressure onset for static ionisation contrasts with the extreme pressures required in the pure ices, and can be understood by the lower energy cost for the proton transfer between H2O and NH3. Future research will investigate the presence of a superionic phase with large proton mobility, which could exist in AMH at milder P-T conditions than in the pure ices (P > 50 GPa, T ~1000 K).
Principal publication and authors
Topologically frustrated ionization in a water-ammonia mixture, C. Liu (a, b), A. Mafety (a), J.A. Queyroux (a), C. W. Wilson (c), H. Zhang (a), K. Béneut (a), G. Le Marchand (a), B. Baptiste (a), P. Dumas (d), G. Garbarino (e), F. Finocchi (f), J.S. Loveday (c), F. Pietrucci (a), A.M. Saitta (a), F. Datchi (a) and S. Ninet (a), Nat. Commun. 8, 1065 (2017); doi:10.1038/s41467-017-01132-z.
(a) Institut de Minéralogie, de Physique des Matériaux et de Cosmochimie (IMPMC), Sorbonne Université, Paris (France)
(b) Institute of Atomic and Molecular Physics and State Key Laboratory of Superhard Materials, Jilin University, Changchun (China)
(c) SUPA, School of Physics Astronomy Centre for Science at Extreme Conditions, University of Edinburgh, (UK)
(d) Synchrotron SOLEIL, Gif sur Yvette (France)
(e) ESRF
(f) Institut des Nanosciences de Paris, Sorbonne Universités, Paris (France)



